Contents
Japan Stem Cell para sa Diabetes: Bagong Pag-asa para sa Type 1 at Type 2
Ang Japan ay nangunguna sa pandaigdigang larangan ng regenerative medicine, na nag-aalok ng ilan sa mga pinaka-advanced at mahigpit na kinokontrol na stem cell therapies para sa Diabetes Mellitus (Type 1 at Type 2). Kasunod ng paggawad ng Nobel Prize kay Dr. Shinya Yamanaka para sa kanyang pagtuklas ng induced pluripotent stem cells (iPSCs), pinabilis ng gobyerno ng Japan ang klinikal na aplikasyon ng mga regenerative therapies, na lumikha ng isang natatanging kapaligiran kung saan ang inobasyon ay nakakatugon sa mahigpit na mga protocol sa kaligtasan.
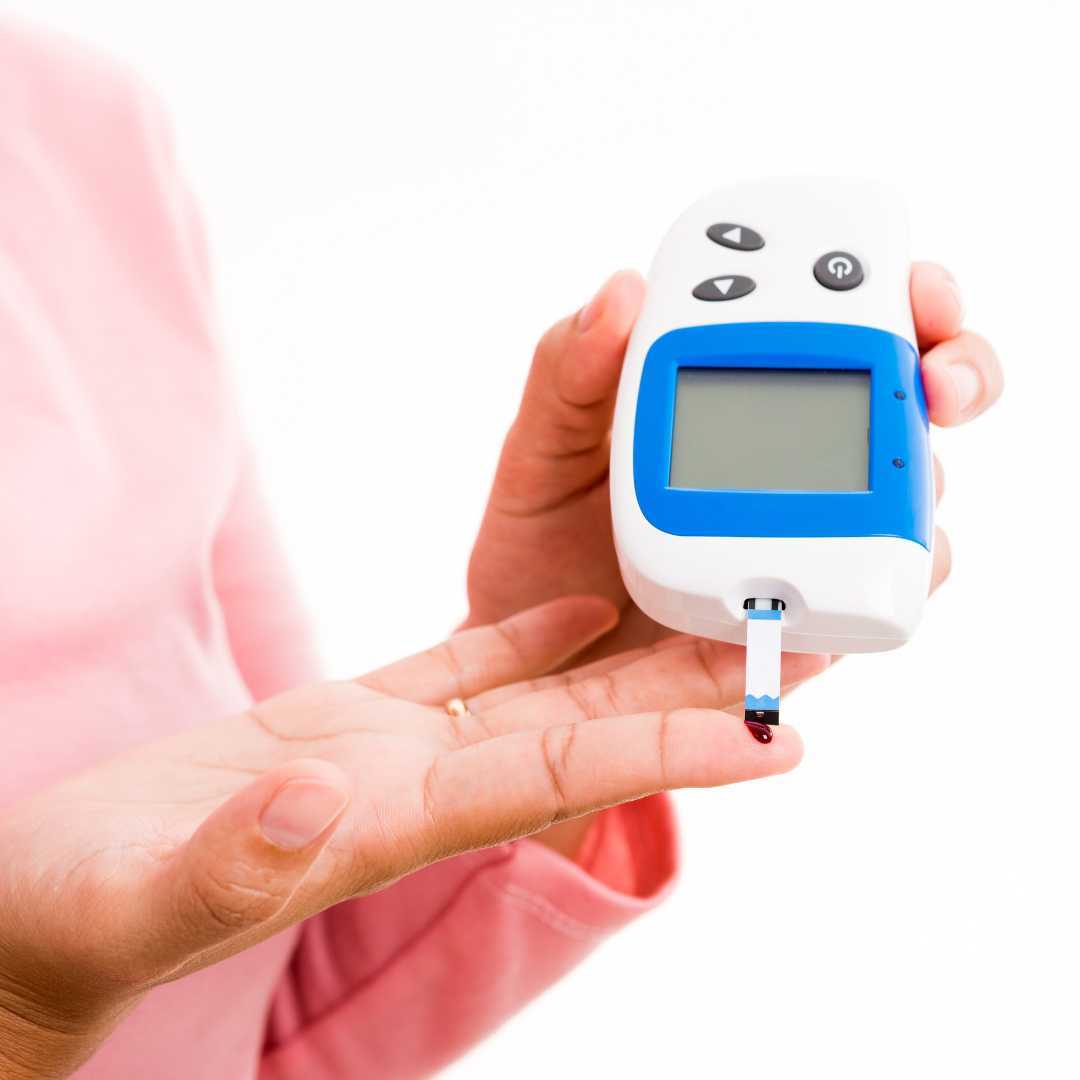
Para sa mga pasyenteng may diabetes, ang paggamot sa Japan ay nakatuon sa pag-aayos ng function ng pancreas, pagpapabuti ng sensitivity ng insulin, at pamamahala ng malulubhang komplikasyon tulad ng neuropathy at nephropathy. Hindi tulad ng maraming iba pang hurisdiksyon kung saan ang mga stem cell clinic ay nagpapatakbo sa isang "gray area," ipinapatupad ng Japan ang Act on the Safety of Regenerative Medicine (ASRM) . Tinitiyak nito na ang bawat klinika na gumagamot sa mga internasyonal na pasyente ay nasuri at lisensyado ng Ministry of Health, Labour and Welfare.
Ang mga pasyenteng naglalakbay sa mga lungsod tulad ng Tokyo, Osaka, o Kyoto ay maaaring asahan ang isang lubos na siyentipikong pamamaraan. Karaniwang gumagamit ang mga paggamot ng autologous Mesenchymal Stem Cells (MSCs) na nagmula sa adipose tissue, na kinultura sa mga high-tech na laboratoryo upang matiyak ang kadalisayan at bisa bago ibigay.
Ang pagpili sa Japan ay isang pagpipilian para sa kalidad, kaligtasan, at katumpakan. Ito ay isang mainam na destinasyon para sa mga pasyenteng naghahanap ng lehitimo at nakabatay sa ebidensyang regenerative care upang mapamahalaan ang kanilang kondisyon at mapabuti ang kanilang kalidad ng buhay.
Alam Mo Ba?
Ang Japan ang unang bansa sa mundo na nagpabilis ng pag-apruba para sa mga produktong regenerative medicine. Ang "Sakigake Strategy" ng gobyerno ay nagpapahintulot sa mga promising therapy na maaprubahan para sa conditional use nang mas mabilis kaysa sa US o Europe, na nagbibigay sa mga pasyente ng mas maagang access sa mga breakthrough treatment sa ilalim ng mahigpit na pagsubaybay.
Mga Pangunahing Pananaw sa Isang Sulyap
Ang mga klinika ay dapat may hawak na lisensyang Class I, II, o III sa ilalim ng Act on the Safety of Regenerative Medicine upang legal na makapagpatakbo.
Ang mga therapy ay lubos na epektibo sa pagtugon sa diabetic neuropathy, mga problema sa paggana ng bato, at kalusugan ng mga ugat.
Gumagamit ang mga laboratoryo sa Hapon ng makabagong teknolohiya sa cell culture (CPC) upang mapalawak ang mga stem cell sa maraming bilang nang hindi nawawala ang bisa.
Kadalasang pinagsasama ng paggamot ang mga stem cell infusions na may gabay sa nutrisyon at mga advanced na diagnostic.
Ang mga internasyonal na pasyente ay karaniwang nangangailangan ng Medical Visa, na nagsisiguro na sila ay ginagamot sa mga akreditadong pasilidad.
Gumagamit ang mga klinika ng mga makabagong pamamaraan ng paghahatid, kabilang ang IV infusion at localized injections, na ginagabayan ng precision imaging.
Ang Tahanan ng mga iPS Cell
Ang Japan ang lugar ng kapanganakan ng Induced Pluripotent Stem Cells (iPS cells) , na natuklasan ni Dr. Shinya Yamanaka ng Kyoto University noong 2006. Bagama't karamihan sa mga kasalukuyang klinikal na paggamot ay gumagamit ng mga adult stem cell (MSC), ang Japan ay aktibong nagsasagawa ng mga pinaka-advanced na klinikal na pagsubok sa mundo gamit ang mga iPS cell upang lumikha ng mga beta cell na gumagawa ng insulin, na naglalapit sa atin sa isang potensyal na lunas para sa Type 1 Diabetes.
Komprehensibo at detalyado ang pangangalagang medikal sa Japan. Karaniwang kasama sa mga pakete ng mga provider na nag-aalok ng stem cell culture process, administration, malawakang pre-treatment testing (blood work, imaging), at post-treatment monitoring ang mga pasyenteng nangangailangan ng pangangalagang medikal sa Japan. Dahil sa mga hadlang sa wika, kasama rin sa mga pinakamahusay na pakete ang isang dedikadong medical interpreter at tulong sa proseso ng aplikasyon para sa Medical Visa (Certificate of Eligibility).
Paalala: Karamihan sa mga pakete ay walang kasamang mga flight o akomodasyon, ngunit maaaring magrekomenda ang mga klinika ng mga kalapit na partner hotel.
Ang Japan ay isang premium na destinasyong medikal, at ang mga gastos ay sumasalamin sa mataas na pamantayan ng pagproseso sa laboratoryo at pagsunod sa kaligtasan. Ang karaniwang gastos para sa isang kurso ng stem cell therapy para sa diabetes ay karaniwang mula $15,000 hanggang $25,000 USD . Ang presyong ito ay kadalasang sumasaklaw sa pag-aani ng adipose tissue, cell culturing (na tumatagal ng 3-4 na linggo), at ang infusion session. Bagama't mas mataas kaysa sa ibang mga bansa, tinitiyak ng gastos na ang mga cell ay pinoproseso sa isang sertipikadong Cell Processing Center (CPC).
Tip: Palaging itanong kung kasama sa presyo ang gastos sa cell culturing/expansion, dahil ito ang pinakamahal na bahagi ng proseso.
Stem Cell Treatment for Diabetes Centers Cost Comparison in Japan
| Provider | Procedure | Price |
|---|---|---|
| HELENE - Stem Cell Clinic | Stem Cell Treatment for Diabetes, Stem Cell Therapy | $10000 |
Stem Cell Treatment for Diabetes Cost Comparison in Japan
| Country | Procedure | Price |
|---|---|---|
| United States | Stem Cell Treatment for Diabetes, Stem Cell Therapy | $22000 |
Nakapaglista kami ng mga pinaka-kagalang-galang na klinika sa Japan na may hawak na mga balidong lisensya mula sa Ministry of Health, Labour and Welfare. Ang mga sentrong ito ay kadalasang pinamumunuan ng mga akademikong mananaliksik at nagpapatakbo sa loob ng mahigpit na mga alituntunin sa etika. Nagtatampok ang mga ito ng mga on-site o partnered na CPC (Cell Processing Centers) upang matiyak ang pinakamataas na posibilidad na mabuhay ang mga stem cell. Suriin ang mga profile sa ibaba upang suriin ang kanilang mga partikular na lisensya (Class II o Class III) at mga protocol sa paggamot.
Pamantayan: Maghanap ng mga klinika na nagpapakita ng kanilang numero ng abiso na "Provision of Regenerative Medicine" sa kanilang profile.
Nagbibigay ng mahalagang katiyakan ang pakikinig mula sa mga pasyenteng nakaranas na ng sistemang medikal ng Hapon. Tampok sa mga testimonial sa ibaba ang mga internasyonal na pasyente na nagbabahagi ng kanilang mga karanasan sa stem cell therapy sa Japan. Tinatalakay nila ang mga pagpapabuti sa kanilang mga antas ng glucose sa dugo (HbA1c), mga pagbawas sa mga sintomas ng neuropathy, at ang propesyonalismo ng mga medikal na pangkat ng Hapon.
Kabatiran: Madalas na pinupuri ng mga pasyente ang kalinisan, pagiging nasa oras, at detalyadong paliwanag na ibinibigay ng mga doktor na Hapones.
Ang mga review para sa medikal na paggamot sa Japan ay palaging nagbibigay-diin sa kulturang "Omotenashi" (pagiging mabuting pakikitungo). Sa seksyong ito, basahin ang mga beripikadong review na sumasaklaw sa buong paglalakbay ng pasyente—mula sa unang malayuang konsultasyon hanggang sa huling paglabas. Madalas na inirerekomenda ng mga pasyente ang Japan dahil sa pakiramdam ng kaligtasan, mga high-tech na pasilidad, at ang pagiging detalyado ng mga medikal na ulat na ibinibigay pagkatapos ng paggamot.
Tip sa Pagsusuri: Maghanap ng mga review na binabanggit ang pangmatagalang follow-up, dahil kilala ang mga klinikang Hapones sa pagpapanatili ng pakikipag-ugnayan.
How Much Does Stem Cell Treatment for Type 1 Diabetes Cost in Japan?
Stem cell treatment for Type 1 diabetes in Japan typically costs between $15,000 and $40,000 USD for standard protocols. Premium packages, which may include higher cell counts or multiple sessions, can range from $50,000 to over $100,000 depending on the clinic and duration of care. Type 1 diabetes is a challenging autoimmune condition where the body’s immune system attacks the insulin-producing beta cells in the pancreas. For decades, the only solution has been lifelong insulin injections and constant blood sugar monitoring. However, recent advancements in regenerative medicine are offering a new avenue of hope. Japan, a world leader in this field, has become a top destination for patients seeking stem cell therapy for Type 1 diabetes. Unlike conventional treatments that merely manage symptoms, stem cell therapy aims to address the underlying immune dysfunction. By utilizing the body’s own repair mechanisms, this therapy seeks to protect remaining beta cells and potentially improve insulin sensitivity. If you are considering traveling to Japan for this cutting-edge treatment, understanding the costs, legalities, and realistic outcomes is crucial. In this comprehensive guide, we will break down everything you need to know to make an informed decision about your health journey. What is stem cell therapy for Type 1 diabetes? Stem cell therapy for Type 1 diabetes involves the administration of mesenchymal stem cells (MSCs) to modulate the immune system, reduce pancreatic inflammation, and potentially preserve or regenerate insulin-producing beta cells. In Type 1 diabetes, the immune system mistakenly identifies beta cells as threats and destroys them. Stem cell therapy works primarily through "immunomodulation." The introduced stem cells act like peacekeepers, signaling the immune system to calm down and stop attacking the pancreas. This creates a more favorable environment for the body to heal. Additionally, these cells release potent growth factors that improve blood flow and reduce systemic inflammation. While the primary goal is to stop the autoimmune attack, there is also hope that this environment allows for the preservation of whatever beta cell function remains, which can significantly stabilize blood sugar levels and reduce the amount of exogenous insulin a patient needs daily. Why choose Japan for Type 1 diabetes treatment? Japan offers a unique combination of advanced medical technology and strict government regulation under the "Act on the Safety of Regenerative Medicine," ensuring that patients receive safe, high-quality, and legally approved cell therapies. Many countries offer stem cell treatments in regulatory gray areas, but Japan is different. The Japanese government has proactively legislated regenerative medicine, creating a framework where clinics must prove the safety and sterility of their procedures before they can treat patients. This provides a layer of consumer protection that is rare in the medical tourism industry. Furthermore, Japan is the birthplace of induced pluripotent stem cell (iPS) technology (Nobel Prize-winning research). While most clinical treatments currently use adult stem cells (MSCs), this strong research culture permeates the clinical landscape, meaning you are often treated by doctors who are at the forefront of global scientific research. Is stem cell therapy for Type 1 diabetes legal in Japan? Yes, it is legal. Clinics must obtain specific approval from the Ministry of Health, Labour and Welfare (MHLW) to offer stem cell therapies for diabetes, and they are rigorously monitored for compliance and safety. You can verify a clinic's legitimacy by asking for their MHLW notification number. This number proves that their specific treatment plan—in this case, using stem cells for diabetes—has been vetted by a certified committee and registered with the government. This transparency is a huge advantage for international patients. It is important to note that while legal, these treatments are generally considered "self-funded" medical care. This means they are not covered by Japanese national insurance for tourists, nor are they typically covered by insurance companies from the US or Europe, as they are still classified as experimental or elective in those regions. What is the cost of stem cell treatment for Type 1 diabetes in Japan? The cost generally falls between $15,000 and $40,000 USD for a standard single-cycle treatment. Comprehensive packages involving multiple infusions or extended hospital stays can exceed $100,000. The price tag reflects the high cost of operating in Japan's regulated environment. The most expensive component is the cell processing. Your cells must be cultured in a specialized, sterile facility known as a Cell Processing Center (CPC) for several weeks to ensure they are free of contaminants and have multiplied to therapeutic numbers. Here is a breakdown of what you might expect to pay: Treatment Package Estimated Cost (USD) Details Standard Protocol $15,000 - $25,000 One harvesting session, lab culture, and one large IV infusion. Advanced Protocol $30,000 - $50,000 Higher cell counts (300M+), cryopreservation for future use, multiple infusions. Premium / Multi-Stage $90,000 - $150,000 Comprehensive "cure" programs with extensive monitoring, genetic testing, and multiple visits. What types of stem cells are used? The most common cell type used in Japan for Type 1 diabetes is Autologous Adipose-Derived Mesenchymal Stem Cells (ADSCs), taken from the patient's own fat tissue. Doctors prefer ADSCs for several reasons. First, they are easily accessible via a simple mini-liposuction procedure. Second, fat tissue is incredibly rich in mesenchymal stem cells—much more so than bone marrow. Third, because they are "autologous" (your own), there is zero risk of your body rejecting them as a foreign object. While Japan is famous for iPS cells (which can theoretically be turned into new pancreatic beta cells), this specific technology is still largely in the clinical trial phase and not widely available for commercial treatment. ADSCs remain the safest and most established option for current patients. How is the procedure performed? The procedure is a multi-step process: first, a small amount of fat is harvested under local anesthesia; next, the cells are cultured in a lab for 3-4 weeks; finally, the expanded cells are returned to the patient via an intravenous (IV) drip. The process is designed to be minimally invasive. On your first visit, the doctor will perform a small liposuction on your abdomen or thigh. This takes less than an hour. You are then free to leave, though you will have to wait a few weeks for the laboratory to do its work. This culture phase is critical; it turns a few thousand cells into hundreds of millions. When you return for the infusion, it is a simple outpatient appointment. The cells are dripped into your bloodstream over the course of 1-2 hours. The MSCs then circulate through the body, homing in on areas of inflammation, including the pancreas. What success rates can I expect? Success is measured by a reduction in insulin requirements and improved HbA1c levels, not necessarily a total cure. Many patients report a "honeymoon period" where blood sugar stabilizes and insulin needs drop by 30-50%. It is vital to manage expectations. Stem cell therapy is not a magic wand that will instantly regrow a full new pancreas. For Type 1 diabetics, the success is often defined by a reduction in the "brittleness" of their diabetes. This means fewer dangerous lows (hypoglycemia) and fewer spikes. Some patients may find they can reduce their daily insulin dosage significantly, while a small percentage of early-stage patients (those diagnosed very recently) may achieve temporary insulin independence. However, for long-term diabetics, the goal is usually stability and the prevention of complications like neuropathy or kidney failure. Can this therapy cure Type 1 Diabetes? Currently, stem cell therapy is considered a treatment, not a guaranteed cure. While it can significantly improve quality of life and reduce symptoms, most patients will still need to use some insulin, albeit often at lower doses. The word "cure" implies that you can walk away and never think about diabetes again. Science is getting closer, but we aren't quite there yet with commercially available treatments. The autoimmune nature of Type 1 diabetes means that even if new beta cells are regenerated, the body may eventually attack them again without ongoing immunomodulation. Think of this therapy as a powerful tool to reset your immune system and preserve your body's remaining function, buying you time and health while protecting your organs from the damage of high blood sugar. What are the risks and side effects? Side effects are generally mild and rare. They may include temporary pain or bruising at the liposuction site, and occasionally a low-grade fever or fatigue for 24 hours after the cell infusion. Because the cells are your own, there is no risk of graft-vs-host disease or rejection, which are major risks in organ transplants. The biggest safety concern in cell therapy is contamination, but Japan's strict regulations on cell processing centers make this risk extremely low. Long-term risks are minimal, but as with any medical procedure, it is important to discuss your full medical history with the doctor to ensure there are no contraindications, such as active cancer or severe infections. What is the recovery time? Recovery is minimal. Patients can typically walk out of the clinic immediately after both the fat harvest and the infusion. Most return to normal non-strenuous daily activities the next day. You won't need to stay in a hospital bed. The liposuction site might feel tender, like a muscle workout or a bruise, for about a week. Doctors usually advise avoiding heavy lifting, intense gym sessions, or swimming/baths for a week to ensure the harvest site heals perfectly. For the infusion, you might feel a bit tired afterwards, so planning a quiet evening at your hotel is a good idea. By the next morning, most patients feel back to normal and are ready to enjoy their time in Japan. Who is eligible for this treatment? Eligible patients are typically those with Type 1 diabetes who are in stable health and free of active infections or cancer. It is often most effective for patients diagnosed more recently who still have some beta cell function (detectable C-peptide). Doctors will ask for your recent blood tests, specifically your HbA1c and C-peptide levels. C-peptide is a marker that shows if your body is still making any of its own insulin. Patients with detectable C-peptide tend to respond better because there are still beta cells left to protect and stimulate. However, even long-term patients with no C-peptide can benefit from the anti-inflammatory effects, which can help treat or prevent complications like diabetic foot, kidney issues, or eye problems. How does this compare to an insulin pump or islet transplant? An insulin pump is a mechanical management tool, while stem cell therapy is a biological repair treatment. Islet transplants are effective but require lifelong immunosuppressant drugs, whereas stem cell therapy uses your own cells and requires no anti-rejection medication. Insulin pumps are fantastic for management, but they do not treat the disease itself. Islet transplants (taking cells from a donor) can cure diabetes, but they trade one disease for another: you must take powerful drugs to suppress your immune system for the rest of your life, which carries high risks. Stem cell therapy attempts to find a middle ground—biological repair without the need for dangerous immunosuppressive drugs. It is a safer, albeit currently less "curative," option compared to a full transplant. Are there specific clinics in Japan for this? Yes, several licensed clinics in Tokyo, Osaka, and Kyoto specialize in diabetes treatment. Notable names often include clinics or those affiliated with major research universities offering private care. When searching for a clinic, look for one that specifically lists "diabetes" in their MHLW-approved provision plan. Some clinics specialize in orthopedic (joint) stem cells, which is a different protocol. You want a clinic that understands the metabolic and autoimmune nature of diabetes. These clinics are often high-end, boutique medical facilities designed to cater to international clientele, providing a comfortable and private environment for treatment. Will I face a language barrier? Leading clinics dealing with international patients typically have English-speaking staff or provide professional medical interpreters to ensure seamless communication during consultations and procedures. Japan has invested heavily in medical tourism infrastructure. You will likely be assigned a dedicated case manager who speaks English. They will handle your appointments, translate your medical documents, and be by your side during the doctor's consultation. Always confirm the availability of interpretation services before you book. Miscommunication in medical settings can be stressful, so having that guaranteed support is worth checking for. How do I get started? The first step is to contact a clinic or a medical tourism agency to submit your medical records. You will need to provide recent blood work (HbA1c, C-peptide, etc.) for a preliminary review by the Japanese medical team. Do not book your flight just yet. You need to be "accepted" as a patient first. This remote consultation ensures that you are a good candidate and that the treatment is likely to help you. Once approved, the clinic will help you coordinate your travel dates. Remember to plan for the timeline: you will either need a long stay (4-5 weeks) or two short trips (3-4 days each) separated by a month. Factor this travel time and cost into your overall budget. Take the Next Step for Your Health If you are ready to explore the potential of stem cell therapy for Type 1 diabetes and want to connect with licensed, safe clinics in Japan, PlacidWay is here to assist you. We simplify the process of finding top-tier medical care abroad.
Is Stem Cell Treatment for Type 1 Diabetes Available in Japan?
Yes, stem cell treatment for type 1 diabetes is available in Japan through highly regulated regenerative medicine frameworks. While still largely in clinical trial or experimental phases, international patients can access specialized therapies. Key Takeaways: Regenerative Diabetes Care Regulatory Excellence: Procedures are strictly governed by the Act on the Safety of Regenerative Medicine. Dual Approach: Therapies focus on both immune system modulation and pancreatic beta cell preservation. Global Accessibility: International patients are welcomed through specialized medical coordination pathways. Cost Considerations: Total financial investment reflects advanced laboratory cell expansion and medical supervision. Scientific Innovation: The nation leads globally in induced pluripotent and mesenchymal cell research. Living with an autoimmune condition requires constant vigilance, and many patients seek innovative solutions beyond daily insulin injections. Over the last decade, regenerative medicine has emerged as a beacon of hope. Asia, particularly the eastern region, has established itself as a global powerhouse in biological research. This comprehensive guide explores how international patients are accessing advanced protocols, understanding the financial requirements, and navigating the healthcare ecosystem to find a reliable Stem Cell Treatment for Type 1 Diabetes in Japan. How does stem cell therapy work for Type 1 Diabetes? Stem cell therapy aims to replace destroyed insulin-producing beta cells and modulate the immune system to stop further attacks. This dual approach helps restore natural glucose regulation in the body. Type 1 diabetes is fundamentally an autoimmune disorder where the body mistakenly attacks its own pancreas. Specifically, the immune system targets and destroys beta cells, which are entirely responsible for producing insulin. Without insulin, glucose cannot enter the cells to provide energy, leading to dangerous blood sugar spikes. Regenerative therapy approaches this complex problem from two distinct angles rather than just managing the symptoms. The first mechanism of action is immunomodulation. When specialized biological cells are introduced into the bloodstream, they travel to areas of high inflammation. Once there, they release anti-inflammatory cytokines that essentially calm the overactive immune system. By halting the autoimmune attack, the therapy protects any remaining healthy beta cells from being destroyed, which is particularly crucial for recently diagnosed patients who are still in their honeymoon phase. The second mechanism involves cellular regeneration and support. While completely regrowing a damaged pancreas is still under heavy research, the introduced cells create a highly supportive microenvironment. They stimulate angiogenesis, which is the formation of new blood vessels, ensuring the damaged pancreatic tissue receives maximum oxygen and nutrients. This supportive environment encourages the body to heal itself and improves the overall efficiency of natural insulin secretion. Expert Insight The homing effect is one of the most remarkable features of mesenchymal cells. When administered intravenously, these cells act like microscopic paramedics. They detect chemical distress signals emitted by the inflamed pancreas and naturally migrate exactly to where the tissue damage is most severe. What types of stem cells are used for diabetes treatment in Japan? Japanese medical facilities primarily utilize Mesenchymal Stem Cells and induced Pluripotent Stem Cells for diabetes management. These cells possess strong regenerative and immunomodulatory properties. The scientific community categorizes biological material based on its origin and potential to transform into other tissue types. In modern Asian healthcare protocols, Mesenchymal Stem Cells are currently the most widely utilized option for autoimmune conditions. These multipotent cells are typically harvested from ethical sources such as umbilical cord tissue, adipose fat tissue, or bone marrow. Because they lack certain cell surface markers, they have an incredibly low risk of being rejected by the patient immune system. Umbilical cord-derived cells are particularly favored in advanced clinics. Unlike autologous cells extracted directly from the patient, cord tissue cells are completely naive to the patient autoimmune disease. If a facility extracts fat cells from a diabetic patient, those cells may already carry genetic predispositions or be fatigued by chronic illness. Cord cells, however, offer pure, youthful vitality and robust multiplication capabilities within the laboratory setting. Looking toward the future, the nation is the undisputed global pioneer in induced Pluripotent Stem Cell technology, following groundbreaking Nobel Prize-winning research. Scientists can take ordinary adult skin cells and genetically reprogram them back into an embryonic-like state. While still largely confined to highly controlled clinical trials, these reprogrammed cells hold the ultimate potential to be transformed precisely into brand new, functional pancreatic beta cells. Are international patients eligible for these regenerative therapies? Yes, foreign nationals can access Stem Cell Treatment for Type 1 Diabetes in Japan if they meet specific medical criteria. Strict pre-screening ensures patient safety and treatment viability. The healthcare system is incredibly welcoming to international patients, provided they follow the rigorous clinical pathways designed for safety. Eligibility is not based on nationality, but rather on highly specific medical parameters. Before a patient is approved to travel, they must undergo a comprehensive remote medical consultation. During this phase, specialists review extensive medical history, current endocrinology reports, and recent laboratory blood panels. One of the most critical eligibility markers is the C-peptide blood test. C-peptide is a byproduct created when the body naturally produces insulin. By measuring this protein, doctors can determine exactly how much residual beta cell function the patient still possesses. Patients who have been recently diagnosed and still have measurable C-peptide levels generally experience the most profound benefits from regenerative immunomodulation. Furthermore, patients must be free of active infectious diseases, severe cardiovascular complications, and advanced kidney failure. Because the therapy modulates the immune system, individuals with active cancer or a history of malignant tumors are generally disqualified to prevent any risk of cellular misbehavior. Psychological readiness and realistic expectations regarding the outcome are also carefully evaluated during the screening process. Did You Know? The local government mandates that every single regenerative medicine protocol must be reviewed and approved by an independent certified committee before it can be offered to the public, ensuring world-class safety standards for every international visitor. What is the average Stem Cell Treatment for Type 1 Diabetes cost in Japan? The Stem Cell Treatment for Type 1 Diabetes cost in Japan generally ranges between $15,000 and $35,000 depending on the protocol. This price reflects the advanced laboratory cultivation and specialized medical care required. Understanding the financial commitment is crucial for anyone planning a medical journey abroad. The total expense is significantly influenced by the complexity of the cellular laboratory work. Unlike generic pharmaceutical drugs that are mass-produced on an assembly line, biological therapies require highly personalized, labor-intensive cultivation. Scientists must isolate, purify, and expand the cells in multi-million dollar cleanroom facilities over several weeks. A comprehensive treatment package typically includes several distinct cost centers. The bulk of the expense covers the cell harvesting and expansion process. Secondary costs include comprehensive diagnostic imaging, baseline metabolic blood testing, physician administration fees, and specialized nursing care during the infusion. Premium medical packages may also bundle airport transfers, dedicated medical interpreters, and postoperative nutritional supplements. While the initial price tag may seem substantial, patients often view this as a long term investment in their health. Uncontrolled diabetes can lead to devastating secondary complications requiring kidney dialysis, vision treatments, or peripheral neuropathy management. By successfully modulating the disease progression early on, patients may significantly reduce their lifetime expenditure on emergency interventions and intensive chronic care management. How do I find a regulated Stem Cell Treatment for Type 1 Diabetes clinic in Japan? Locating a certified Stem Cell Treatment for Type 1 Diabetes clinic in Japan requires checking the facility against the strict registry of the Ministry of Health, Labour and Welfare. Only licensed facilities are legally permitted to offer these protocols. Because regenerative medicine is a highly specialized field, patients must exercise extreme diligence when selecting a healthcare provider. The national regulatory framework is among the strictest in the world, which is excellent for patient safety but requires careful navigation. Here is how you identify a premier medical facility: Government Certification: Verify that the facility holds an active license under the Act on the Safety of Regenerative Medicine. In-House Cultivation: Top tier facilities operate their own Cell Processing Centers to ensure absolute quality control without relying on third party transport. Specialized Medical Teams: Look for departments headed by board-certified endocrinologists who work alongside expert immunologists. Transparent Protocols: Reputable centers will clearly document their exact cell count dosages and administration methodology. International Patient Departments: Dedicated multilingual support teams indicate the facility has robust experience handling foreign medical records. What are the expected results after receiving this treatment? Patients often experience improved HbA1c levels, reduced daily insulin requirements, and fewer hypoglycemic episodes. Results vary based on the duration of the disease and individual metabolic responses. It is vital to approach regenerative therapies with medically accurate expectations. While complete independence from insulin is the ultimate holy grail of endocrinology research, current practical results focus on deep disease management. Many patients report a stabilization of their blood sugar fluctuations within the first three to six months following the cellular infusion, leading to a profound improvement in daily quality of life. A primary clinical marker of success is a gradual reduction in the required daily units of exogenous insulin. As the introduced cells lower systemic inflammation and encourage whatever natural pancreatic function remains, the body becomes more efficient. Patients frequently observe a downward trend in their HbA1c readings, which is the gold standard measurement for long term glucose management. Beyond the numbers on a glucometer, patients often highlight subjective health improvements. Enhanced natural energy levels, better sleep quality, and a significant reduction in terrifying sudden hypoglycemic drops are commonly reported. By smoothing out the drastic peaks and valleys of blood sugar levels, the therapy acts as a powerful preventative measure against long term organ damage. Are there any risks associated with this regenerative procedure? While highly monitored, potential risks include minor infection at the injection site, transient fever, or immune rejection. Japan mitigates these risks through exceptionally stringent clinical protocols and laboratory standards. All advanced medical interventions carry inherent risks, and biological therapy is no exception. However, thanks to rigorous scientific advancement, the severe risks associated with early experimental treatments have been largely eliminated. The most common immediate side effects are incredibly mild and typically consist of fatigue, a low grade fever, or slight soreness where the intravenous line was placed. In the past, there were theoretical concerns about cellular mutations or uncontrolled cell growth. Modern regulatory frameworks solve this by mandating extensive chromosomal screening of all biological material before it ever enters a patient. Highly controlled laboratory environments ensure that the cells are stable, pure, and entirely free from bacterial or viral contamination. The risk of immune rejection is heavily mitigated by the intelligent selection of cell types. Because mesenchymal cells lack human leukocyte antigens on their surface, they effectively fly under the radar of the patient immune system. Nonetheless, continuous medical supervision during and immediately after the procedure ensures that any rare hypersensitivity reactions can be managed instantly by the clinical team. How does medical tourism in Japan support patients seeking diabetes care? Medical tourism in Japan offers dedicated infrastructure including specialized medical visas, multilingual support staff, and streamlined clinical pathways. This ecosystem ensures international patients receive seamless healthcare delivery. The nation has actively cultivated a highly sophisticated medical travel infrastructure designed to attract patients seeking world class technological interventions. Recognizing the immense global demand for advanced cellular therapies, the government created the specialized Medical Stay Visa. This visa allows international patients and their accompanying family members to remain in the country for extended periods to complete complex treatment protocols without typical tourist restrictions. Language barriers are a common concern, but the modern healthcare ecosystem has addressed this elegantly. Major international hospitals employ certified medical interpreters who are fluent in complex biological terminology. These professionals remain by the patient side during all consultations, ensuring that informed consent is crystal clear and that patients feel entirely comfortable discussing nuanced health details with their physicians. Furthermore, clinical coordination agencies provide end to end logistics management. From translating hometown medical records before arrival, to scheduling sophisticated laboratory tests in a specific sequence, the entire journey is mapped out precisely. This highly organized approach eliminates the stress of navigating a foreign healthcare system, allowing the patient to focus solely on their rest and cellular recovery. How long do I need to stay in the country for the procedure and recovery? Most international patients should plan to stay for approximately two to three weeks to complete the necessary consultations, the cell infusion, and initial follow-up. This timeline ensures any immediate post-treatment observations are handled properly. A successful regenerative procedure requires patience and precise scheduling. Unlike simple outpatient surgeries, cellular therapy operates on a biological timeline. The first few days of the trip are dedicated to comprehensive health screenings. Doctors will draw fresh blood to confirm the patient baseline metrics match the translated records sent months prior. Any slight discrepancy in kidney function or inflammation markers must be addressed before proceeding. If the protocol utilizes autologous cells, a minor extraction procedure will take place, followed by a waiting period while the laboratory cultivates the biological material. If donor cells are utilized, the infusion can happen much sooner. The actual administration day is surprisingly brief, but the patient must remain near the facility for the critical observation period to monitor for any delayed physiological responses. The final phase of the stay involves extensive postoperative consultations. The endocrinologist will review early systemic reactions and formulate a detailed long term strategy. Patients are provided with a thick dossier of translated medical reports, specific dietary guidelines, and precise instructions for their hometown physician. Rushing back to the airport too soon is highly discouraged to ensure absolute patient safety. Why Choose PlacidWay for your medical journey? PlacidWay connects you with globally accredited healthcare providers while ensuring complete transparency and comprehensive patient advocacy. Our platform simplifies the process of accessing complex international medical care. Attempting to arrange highly complex cellular treatments independently in a foreign language can be incredibly overwhelming. PlacidWay serves as your dedicated healthcare advocate, ensuring safety and clarity at every step. Here is why thousands trust our platform: Rigorous Facility Vetting: We partner exclusively with hospitals and laboratories that comply with stringent national regenerative medicine laws and maintain impeccable safety records. Complete Price Transparency: We negotiate comprehensive medical packages so you understand the exact laboratory, physician, and facility fees long before you make any financial commitment. Dedicated Patient Advocacy: Our team acts as an intermediary, ensuring all your questions are answered clearly by the clinical staff without any medical terminology confusion. Seamless Record Transfer: We facilitate the secure exchange of your complex endocrinology files, guaranteeing the foreign specialists have total insight into your medical history. Uncompromising Ethical Standards: We only align with providers who utilize ethically sourced biological materials and refuse to promote unproven or highly experimental procedures without regulatory oversight. Frequently Asked Questions About Regenerative Diabetes Care Does insurance cover stem cell therapy for diabetes in Japan? Because this is an advanced regenerative procedure, standard international health insurance and domestic national health insurance plans typically do not cover the expenses. Patients usually fund this procedure out of pocket. It is highly recommended to check with private healthcare providers for specialized international coverage options. At what age can a patient receive stem cell therapy for diabetes? Most certified facilities require patients to be adults over the age of eighteen to undergo regenerative protocols. Pediatric cases are exceptionally rare and generally restricted to highly specific clinical trials with strict ethical board oversight. Age limits ensure the immune system is fully developed before modulation. Can stem cells completely cure type 1 diabetes? At this current stage of medical science, this procedure is considered a highly advanced management tool rather than an absolute cure. The goal is to significantly reduce insulin dependency and protect against long term diabetic complications. Ongoing research aims to eventually achieve complete independence from external insulin. How long does a typical stem cell infusion session take? The actual intravenous administration of the biological material is surprisingly quick, often taking between one and three hours. However, the patient will spend several hours at the facility for pre-infusion vital checks and post-infusion observation. The entire daily hospital visit usually spans a half day. Do I need to stop taking insulin before the procedure? Patients must never abruptly stop their prescribed insulin regimen without direct orders from their attending endocrinologist. The medical team will provide a highly specific medication schedule leading up to the procedure day. Insulin doses are gradually adjusted based on post-treatment glucose monitoring results. Is umbilical cord blood used in Japanese diabetes treatments? Wharton jelly derived from umbilical cords is frequently utilized because it contains an abundance of highly potent mesenchymal cells. These ethical and non-controversial sources are prized for their youthful vitality and low risk of immune rejection. All cord tissue is rigorously screened according to strict national biological safety laws. Are translators provided during medical consultations in Japanese clinics? Top tier international healthcare facilities typically employ dedicated in-house medical interpreters who specialize in complex endocrinology terminology. These professionals ensure that all clinical instructions, risks, and expected outcomes are perfectly understood by the patient. Effective communication is a legally mandated part of informed consent. What dietary restrictions exist after stem cell therapy for diabetes? Following the procedure, patients are strongly advised to adhere to an anti-inflammatory diet rich in antioxidants to support cellular regeneration. Excessive alcohol consumption, highly processed sugars, and smoking must be strictly avoided as they can destroy the newly introduced cells. Nutritional counseling is often provided as part of the holistic aftercare program. How soon can I fly back home after receiving stem cell injections? Aviation travel is generally permitted three to five days after the final cellular infusion, provided the patient passes a clearance examination. The short delay helps doctors monitor for any delayed minor reactions like low grade fever. Patients are given comprehensive documentation to carry during their flight. Will my home country doctor be able to track my post-treatment progress? International healthcare facilities provide detailed discharge summaries containing all laboratory reports, cell count data, and follow-up protocols. Your local endocrinologist will receive precise guidelines on how to monitor your c-peptide levels and adjust your insulin over the coming months. Collaborative care ensures long term success. Discover Innovative Diabetes Care With PlacidWay Take control of your autoimmune health by exploring world-class regenerative options. PlacidWay connects you directly with strictly verified, highly advanced medical centers. Submit your medical details today to receive a personalized, no-obligation evaluation from top international endocrinology experts. Get Your Free Quote
Ang paggamot sa Japan ay pinangungunahan ng mga clinician-scientist na kadalasang nasa makabagong larangan ng regenerative research. Nagtatampok kami ng mga espesyalistang lisensyado sa ilalim ng ASRM at may malawak na karanasan sa mga sakit na metabolic. Tinitiyak ng kanilang kadalubhasaan na ang iyong protocol sa paggamot ay batay sa pinakabagong ebidensyang siyentipiko at iniayon sa iyong mga partikular na pangangailangan sa kalusugan.
Mga Sertipikadong Regenerative na Doktor
May lisensya mula sa Ministri ng Kalusugan
Ang mga nangungunang espesyalista ay dapat sumailalim sa mga partikular na pagsasanay at sertipikasyon upang magbigay ng mga cell therapy. Sila ay mga eksperto hindi lamang sa endocrinology, kundi pati na rin sa ligtas na paghawak at pangangasiwa ng mga produktong biyolohikal.
Mga Pinuno ng Pananaliksik
Kahusayan sa Akademiko at Klinikal
Maraming practitioner ang may hawak na mga posisyon sa mga prestihiyosong unibersidad sa Tokyo at Osaka. Madalas silang nag-aambag sa mga pandaigdigang journal tungkol sa regenerative medicine, tinitiyak na direktang inilalapat nila ang mga pinakabagong natuklasan sa pangangalaga sa pasyente.
Pamamaraan sa Kaligtasan Una
Mahigpit na mga Protokol
Kilala ang mga espesyalistang Hapones sa kanilang konserbatibo at inuuna ang kaligtasan. Nagsasagawa sila ng malawakang pre-screening upang matiyak na ikaw ay isang mahusay na kandidato, inuuna ang iyong pangmatagalang kalusugan kaysa sa mabilisang lunas.
Regulasyon sa Pamantayang Ginto
Ang pangunahing dahilan kung bakit pinipili ng mga pasyente ang Japan ay ang kapanatagan ng loob. Ang Act on the Safety of Regenerative Medicine (ASRM) ay nagbibigay ng isang malinaw na balangkas ng batas na nag-aalis ng mga "cowboy clinic."
Alam ng mga pasyente na ang anumang klinika na kanilang binibisita ay sinuri na ng mga komiteng inaprubahan ng gobyerno, na tinitiyak ang pinakamataas na pamantayan ng etika at kaligtasan sa mundo.
Mas Mahusay na Pagproseso ng Selula
Ang kalidad ng mga stem cell ang nagtatakda ng tagumpay ng paggamot. Ang mga Cell Processing Center (CPC) ng Japan ay makabago, na gumagamit ng mga advanced na culture medium (kadalasang walang serum) upang mapalawak ang mga selula sa milyun-milyon nang walang kontaminasyon.
Tinitiyak nito na ang huling produktong ipinapasok sa iyong katawan ay lubos na mabisa at aktibo.
Tiwala at Serbisyong Pangkultura
Kilala ang pangangalagang pangkalusugan ng Hapon sa masusing atensyon nito sa detalye at respeto sa pasyente. Mula sa tumpak na iskedyul hanggang sa malinis na mga pasilidad, ang buong karanasan ay idinisenyo upang mabawasan ang stress.
Ang suportang kapaligirang ito ay mahalaga para sa mga pasyenteng namamahala ng mga malalang kondisyon tulad ng diabetes.
Ang pag-access sa mas mataas na pangangalagang medikal sa Japan ay maaaring maging mahirap dahil sa mga hadlang sa wika at regulasyon. Ang PlacidWay ay nagsisilbing iyong mapagkakatiwalaang tulay, na nag-uugnay sa iyo sa mga lisensyadong klinika at tinitiyak ang isang maayos at sumusunod na paglalakbay.
Pag-verify ng Lisensya
Kinukumpirma namin na ang bawat kasosyong klinika ay may hawak na balidong lisensya mula sa Japanese Ministry of Health upang magsagawa ng mga stem cell therapy, na ginagarantiyahan ang iyong kaligtasan.
Suporta sa Medikal na Visa
Ikinokonekta ka namin sa mga rehistradong guarantor sa Japan na awtorisadong mag-isyu ng mga dokumentong kinakailangan para sa iyong aplikasyon sa Medical Visa.
Mga Konsultasyon sa Malayuang Lugar
Nagbibigay kami ng mga unang video call kasama ang mga espesyalistang Hapones (kasama ang mga interpreter) upang suriin ang iyong medikal na kasaysayan at matukoy ang iyong pagiging kwalipikado bago ka maglakbay.
Mga Serbisyo ng Tagapagsalin
Tinitiyak namin na kasama sa iyong pakete ang propesyonal na interpretasyong medikal, upang lubos mong maunawaan ang iyong plano sa paggamot at ang mga tagubilin ng doktor.
Transparent na Pagpepresyo
Kumuha ng detalyadong mga quote na nagpapaliwanag ng mga gastos sa cell culture, administrasyon, at mga bayarin sa ospital, na tutulong sa iyong planuhin ang iyong pamumuhunan nang walang mga sorpresa.
Pagtataguyod ng Pasyente
Mula sa iyong unang pagtatanong hanggang sa follow-up pagkatapos ng paggamot, susuportahan ka ng aming koponan, tinitiyak na ang iyong karanasan sa Japan ay komportable, magalang, at epektibo.
Damhin ang kinabukasan ng medisina ngayon. Makipag-ugnayan sa PlacidWay upang makatanggap ng libre at personalized na quote at simulan ang iyong paglalakbay tungo sa mas maayos na kalusugan gamit ang stem cell therapy sa Japan.
Kunin ang Iyong Libreng Personalized na Sipi
.png)







.png)




.png)
.png)




Share this listing