CAR-T Cell Therapy Costs in China: Comprehensive Insights and Global Analysis
When facing a formidable cancer diagnosis, the soaring financial burden of modern treatments often adds devastating stress to an already critical situation. Understanding the CAR-T cell therapy costs in China has become a vital lifeline for countless international patients seeking advanced, life-saving immunotherapy. For years, patients with refractory blood cancers have been priced out of revolutionary treatments in Western nations.
Today, there is a paradigm-shifting alternative. Why mortgage your future for localized care when you can access world-class oncology innovations overseas? Watch to see how cutting-edge biotechnology in Asia is offering affordable cancer treatment abroad without compromising on medical excellence.
Video Chapters & Quick Navigation
The landscape of oncology has been forever altered by the advent of chimeric antigen receptor (CAR) T-cell therapy. For patients navigating the terrifying realities of relapsed or refractory hematological malignancies, this treatment represents not just hope, but a genuine chance at long-term remission. However, the geographic disparity in access to this life-saving treatment is profound. As highlighted in the video at , thousands of patients are now looking beyond their domestic borders to find accessible care.
China has rapidly emerged as a global epicenter for cellular immunotherapy, matching and in some metrics exceeding the clinical trial output of the United States. Medical tourists from North America, Europe, and neighboring Asian countries are traveling to major Chinese metropolises to access innovative cancer treatments. By exploring the deep nuances of CAR-T cell therapy costs in China, patients and their families can make empowered, educated decisions about their healthcare journey.
Understanding the Science: What is CAR-T Cell Therapy?
To fully appreciate the cost dynamics, it is crucial to understand why this treatment is so complex. CAR-T cell therapy is not a traditional pharmaceutical drug that can be mass-produced in a factory. It is a highly personalized, autologous treatment, meaning the medication is created entirely from the patient's own biological material.
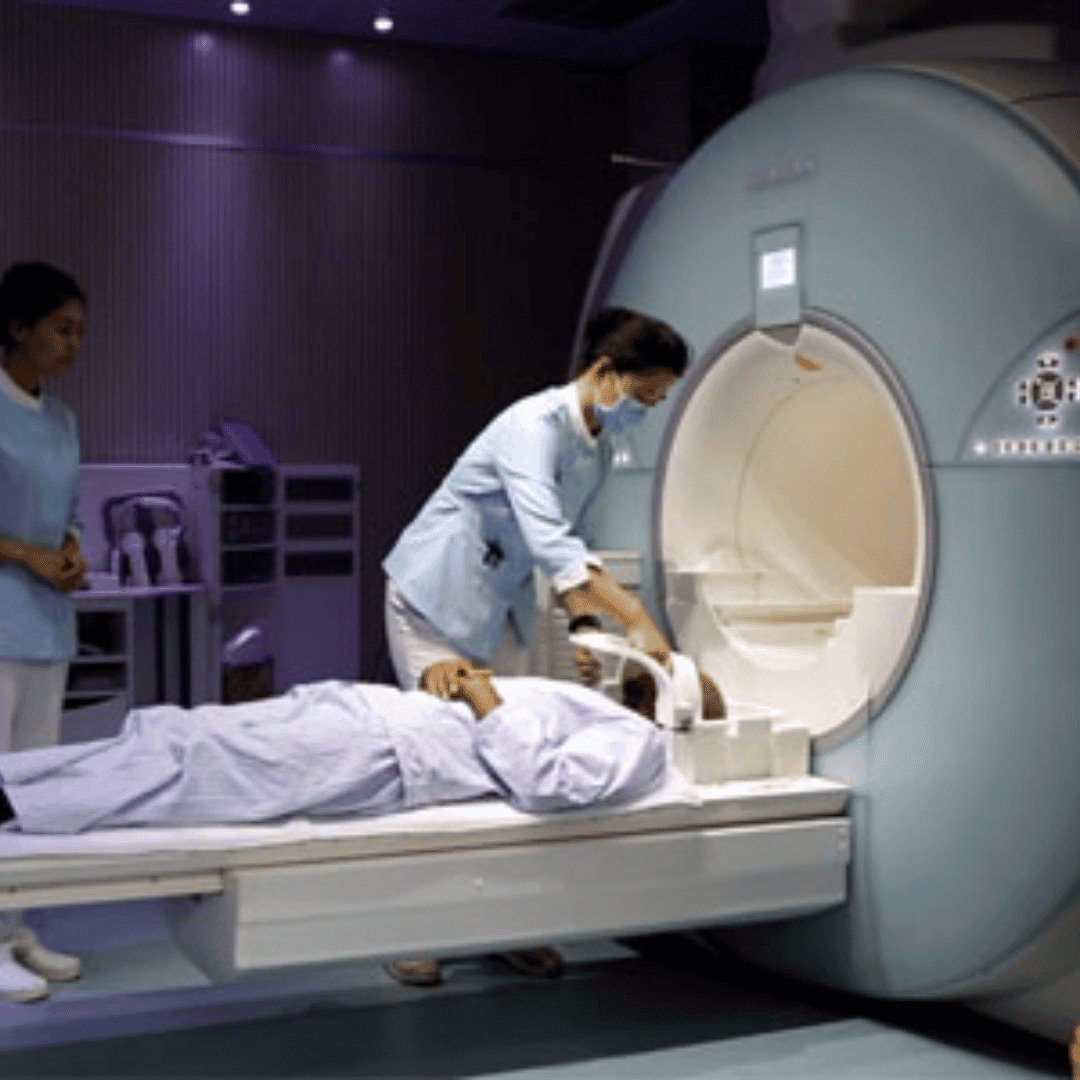
The process begins with leukapheresis, where blood is drawn from the patient to extract T-cells, the immune system's primary warriors. These cells are then transported to a specialized biotechnology laboratory. In the lab, scientists use viral vectors to genetically engineer these T-cells, introducing a chimeric antigen receptor (CAR) that is specifically designed to recognize and bind to unique proteins found on the surface of cancer cells, such as the CD19 antigen.
Once the cells are successfully modified, they are multiplied by the millions. After a brief period of lymphodepleting chemotherapy to prepare the patient's body, the newly engineered "super cells" are infused back into the bloodstream. As discussed at , these cells act as a living drug, hunting down and destroying malignant tumors with astonishing precision. Because the manufacturing process requires sterile environments, highly skilled geneticists, and strict regulatory oversight, the baseline cost of production is inherently high.
The Global Financial Burden of Cancer Immunotherapy
The pricing of CAR-T cell therapy in Western nations has sparked fierce debates regarding healthcare equity and patient accessibility. In the United States, FDA-approved CAR-T therapies like Kymriah (tisagenlecleucel) and Yescarta (axicabtagene ciloleucel) carry astronomical price tags. The list price for the cellular product alone typically ranges from $373,000 to over $475,000.
However, the total financial burden is vastly larger. When factoring in the costs of leukapheresis, bridging chemotherapy, hospital stays in intensive care units (ICU), and the management of severe side effects, the total billed cost for an American patient can easily exceed $1 million. Even with robust health insurance, deductibles and out-of-pocket maximums can bankrupt families already struggling with a cancer diagnosis.
European healthcare systems face similar struggles, often leading to strict rationing of the therapy. National health services may only approve the treatment for very specific, narrow demographics, leaving many patients without domestic options. This systemic financial barrier has forced a critical mass of patients to seek affordable cancer treatment abroad, shifting the focus eastward to nations with burgeoning biotech sectors.
Analyzing CAR-T Cell Therapy Costs in China
The financial narrative changes dramatically when examining the medical landscape in Asia. The cost of CAR T-cell therapy in China is a fraction of its Western counterparts, making it a highly attractive destination for medical tourism. How much exactly does it cost? While prices vary based on the specific hospital and the targeted antigen, comprehensive treatment packages generally range from $35,000 to $70,000.
This dramatic reduction in price is not a reflection of compromised quality, but rather a unique combination of governmental subsidies, lower labor costs for skilled scientists, and an intensely competitive domestic biotechnology market. As noted in the video at , China has designated biotechnology as a key strategic pillar of its national development plan, pouring billions of dollars into research and development to lower the cost of raw materials and viral vectors.
Cost Comparison Breakdown
To illustrate the stark differences in pricing, consider the following comparative data. This table highlights the general estimates for total treatment cycles, including hospital stays and standard medical observation.
| Treatment Component | United States Estimated Cost | China Estimated Cost |
|---|---|---|
| CAR-T Cell Manufacturing (Drug Price) | $375,000 - $475,000 | $30,000 - $50,000 |
| Hospital Stay & Administration | $150,000 - $300,000 | $5,000 - $15,000 |
| Side Effect Management (e.g., ICU for CRS) | $50,000 - $250,000 | $2,000 - $8,000 |
| Total Estimated Patient Burden | $575,000 - $1,025,000+ | $37,000 - $73,000 |
Why China is Becoming a Global Hub for Immunotherapy
The affordability of treatment is only one piece of the puzzle. The true driver of medical tourism for cancer treatment in Asia is the sheer volume of clinical innovation occurring within Chinese borders. China currently conducts more clinical trials for CAR-T cell therapy than any other nation in the world, including the United States. This rapid pace of clinical testing allows hospitals to iterate on treatments faster, improving safety profiles and efficacy rates.
Furthermore, the regulatory environment in China, overseen by the National Medical Products Administration (NMPA), has streamlined the approval process for breakthrough therapies while maintaining strict safety and quality standards. This efficiency allows Chinese biotech firms to bring next-generation therapies, such as dual-target CAR-T cells (which target both CD19 and CD22 simultaneously to prevent antigen escape), to the bedside much faster than Western regulatory bodies permit.
By hosting a vast array of clinical trials, many top-tier Chinese oncology centers are able to offer treatments at subsidized rates or even entirely free of charge for qualifying trial participants. As discussed at , this creates unprecedented access to cutting-edge medicine for international patients who have exhausted standard protocols in their home countries.
Types of Cancers Treated with CAR-T in China
While early iterations of CAR-T therapy were strictly limited to specific blood cancers, the scope of treatable conditions in China is rapidly expanding. The innovative immunotherapy options available in major Chinese medical centers encompass a broad spectrum of hematological malignancies, and researchers are making aggressive strides into treating solid tumors.
Patients traveling for medical care will find comprehensive treatment programs targeting the following critical conditions:
- Acute Lymphoblastic Leukemia (ALL): Highly effective in pediatric and young adult populations, achieving remarkable remission rates in patients who have relapsed after stem cell transplants.
- Diffuse Large B-Cell Lymphoma (DLBCL): The most common type of non-Hodgkin lymphoma, treated aggressively with CD19-targeted therapies that have shown excellent durable response rates.
- Multiple Myeloma: Utilizing BCMA (B-cell maturation antigen) targeted CAR-T cells, Chinese clinics are offering robust treatment protocols for patients with refractory multiple myeloma, a condition previously considered incredibly difficult to treat.
- Emerging Solid Tumors: Though still largely in the clinical trial phase, specialized centers are pioneering trials targeting hepatocellular carcinoma (liver cancer), gastric cancer, and pancreatic tumors using advanced mesothelin and claudin18.2 targets.
Leading Hospitals and World-Class Medical Facilities
A common concern among prospective medical tourists is the quality of the hospital infrastructure. It is essential to understand that the facilities administering these advanced cellular therapies in China are state-of-the-art, internationally accredited institutions. Cities like Beijing, Shanghai, Guangzhou, and Shenzhen host massive medical complexes that dwarf many Western hospitals in both size and technological capability.
These leading institutions boast dedicated International Patient Departments (IPDs) designed specifically to cater to foreign visitors. These wings offer VIP suites that resemble luxury hotels, private translation services, and dietary accommodations to ensure patient comfort. The hematology departments are equipped with ultra-clean laminar flow wards, essential for protecting immunocompromised patients during the critical period following their CAR-T cell infusion.
Moreover, the attending oncologists, hematologists, and research scientists leading these programs are often globally educated. Many have completed fellowships or tenures at prestigious institutions like the Mayo Clinic, Johns Hopkins, or MD Anderson Cancer Center before returning to lead departments in China. This cross-pollination of medical knowledge ensures that the standard of care aligns perfectly with international best practices.
Navigating the Logistics of Medical Tourism for Cancer Patients
Traveling halfway across the world for complex cancer treatment is a daunting prospect. It requires meticulous logistical planning, a deep understanding of visa requirements, and precise medical coordination. The journey begins long before the patient boards an airplane. Initial consultations are typically conducted via secure telehealth platforms, where the Chinese oncology team reviews the patient's domestic medical records, bone marrow biopsies, and PET-CT scans to determine initial eligibility.
Once deemed a candidate, the logistical machinery kicks into gear. Patients must secure a medical visa, which the host hospital facilitates by issuing an official letter of invitation. Because cancer treatment cannot wait, expedited visa processing is often available. Upon arrival, the hospital's international concierge team typically meets the patient at the airport, providing private medical transport directly to the facility or a nearby partner hotel.
Language barriers, often perceived as a major hurdle, are mitigated by the presence of dedicated, full-time medical interpreters who accompany the patient to every consultation, scan, and procedure. These interpreters are not just fluent in English; they are trained in complex medical terminology to ensure absolute clarity between the patient and the attending physicians, as highlighted at in the video.
Patient Eligibility and Pre-Treatment Evaluation
Not every cancer patient is an immediate candidate for CAR-T cell therapy. Because the treatment relies on modifying the patient's existing T-cells, the individual must have a sufficient absolute lymphocyte count. Furthermore, patients must be physically resilient enough to withstand the rigorous lymphodepleting chemotherapy and potential post-infusion complications.
The pre-treatment evaluation in China is exhaustive. Upon admission, patients undergo a battery of baseline assessments, including echocardiograms to check heart function, pulmonary function tests, extensive blood panels, and neurological baseline testing. This rigorous screening ensures that the treatment is only administered when the statistical probability of success outweighs the inherent medical risks.
Post-Treatment Care and Side Effect Management
The most critical phase of CAR-T cell therapy is not the infusion itself, but the 30-day monitoring period that follows. As the engineered T-cells multiply rapidly inside the body and begin destroying cancer cells, they release massive amounts of inflammatory cytokines. This triggers a systemic inflammatory response known as Cytokine Release Syndrome (CRS).
CRS can cause high fevers, drastic drops in blood pressure, and respiratory difficulties. Additionally, patients may experience Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS), leading to temporary confusion or aphasia. Chinese oncology centers are world leaders in recognizing and managing these complications. ICUs are primed with tocilizumab (an immunosuppressive drug used specifically to reverse severe CRS) and corticosteroids to rapidly stabilize patients.
Because the cost of prolonged hospital stays in China is significantly lower than in the US, patients are kept under direct medical observation for much longer periods. Instead of being rushed out of the hospital after a few days, international patients typically remain in the step-down unit for weeks, ensuring they only fly home when they are entirely medically stable and firmly on the path to remission.
Long-Term Remission and the Path Forward
The ultimate goal of undertaking this journey is durable, long-term remission. The clinical outcomes from top Chinese institutions mirror the success rates of elite Western hospitals. Many patients with heavily pre-treated, refractory lymphomas and leukemias achieve complete metabolic responses within 30 to 90 days of their infusion. The persistence of the CAR-T cells acts as a long-term surveillance system, guarding against future relapses.
Follow-up care is seamlessly integrated into the medical tourism package. Upon returning to their home countries, patients continue routine blood monitoring and PET scans with their local oncologists. The Chinese medical team maintains open lines of communication with the patient's domestic doctors, creating a collaborative, global approach to long-term cancer survivorship.
The high costs of cancer treatments at home should not be a death sentence. By leveraging the phenomenal advancements and affordable pricing structures available in China, patients can bypass systemic healthcare bottlenecks. It is a profound realization that a flight across the globe could be the definitive step toward reclaiming your health, your future, and your life.
Ready to Explore Lifesaving Immunotherapy Options?
PlacidWay Medical Tourism connects you with globally accredited, top-rated oncology centers in China offering cutting-edge CAR-T cell therapy at a fraction of Western costs. Let our experts guide you through the logistics so you can focus entirely on your healing.
GET A FREE CONSULTATION


.png)



.png)






Share this listing