What treatment
Explore Most Popular Medical Articles Worldwide
Read Top Healthcare Updates
Trusted Medical Tourism Platform Since 2007
Endometriosis Treatment Abroad: Why Patients Choose Tijuana for Specialized Care
Overview
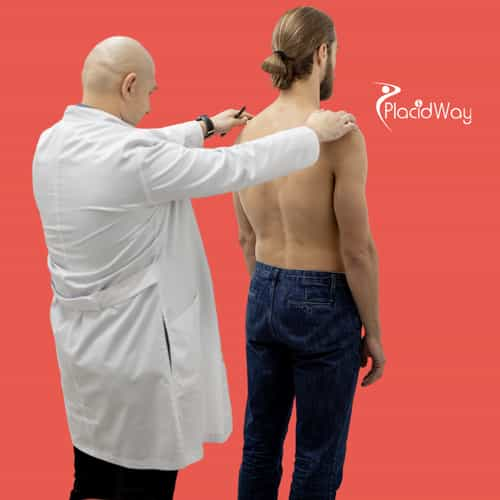
Endometriosis treatment in Tijuana, Mexico, provides patients with access to specialized, minimally invasive surgical care for a chronic condition that often causes severe pelvic pain, infertility, and diminished quality of life. The city’s medical centers feature board-certified gynecologists trained in advanced laparoscopic techniques, offering precise excision of endometrial tissue to improve long-term outcomes. International patients benefit from lower treatment costs, streamlined medical tourism processes, and personalized care coordination, while accredited hospitals maintain high safety and sterility standards comparable to U.S. facilities....
Read more detailsRegaining Weight After Surgery? 5 Signs You Need a Gastric Sleeve Revision in Tijuana, Mexico
Overview
A gastric sleeve revision in Tijuana, Mexico, is a specialized procedure designed for patients experiencing weight regain or unresolved health issues after their initial bariatric surgery. The surgery restores stomach restriction and supports long-term metabolic health, with outcomes strongly influenced by the expertise of board-certified surgeons and adherence to lifestyle changes. Tijuana is a leading medical tourism destination, offering cost-effective care in accredited facilities that follow rigorous safety standards. Patients benefit from structured pre- and post-operative guidance, ensuring a smooth recovery and sustained improvements in weight management and overall health....
Read more detailsEscaping US Medical Debt: Why Hernia Surgery in Mexico is the Best Option
Overview
This article provides a comprehensive overview of hernia surgery in Mexico for international patients seeking affordable, high-quality repair options abroad. It explains common hernia types, including inguinal, umbilical, hiatal, and incisional or ventral hernias, while covering procedure costs, minimally invasive laparoscopic techniques, pre-operative preparation, hospital standards, surgeon selection, recovery guidelines, and cross-border travel logistics. The article highlights Mexico’s role as a major medical tourism destination, emphasizing transparent pricing, shorter wait times, bilingual care teams, accredited facilities, and proper post-operative planning for safer and more organized hernia repair....
Read more detailsBudgeting For Your Medical Trip: Hidden Savings in Tijuana Hip Surgery
Overview
This article provides a comprehensive guide to hip surgery in Tijuana for international patients seeking affordable orthopedic care abroad. It explains common procedures such as total hip replacement, partial hip replacement, hip resurfacing, and revision hip surgery, while also covering treatment costs, pre-operative preparation, recovery timelines, rehabilitation, hospital standards, surgeon selection, and cross-border travel logistics. The article highlights Tijuana’s accessibility for North American patients, its lower surgical costs compared to the US and Canada, and the importance of choosing accredited facilities, experienced orthopedic specialists, and structured post-operative care for a safer medical tourism journey....
Read more detailsComparing Gastric Bypass Costs: Out-of-Pocket Prices in US Hospitals vs Tijuana Mexico
Overview
This article explains the cost, benefits, safety considerations, and patient journey for gastric bypass in Tijuana, Mexico. It highlights why many uninsured or underinsured patients consider Tijuana for affordable bariatric surgery, with typical gastric bypass pricing ranging from $5,500 to $7,500 compared to much higher out-of-pocket costs in the United States. The article also covers package inclusions, surgeon and facility selection, recovery expectations, risks, and how PlacidWay helps patients connect with vetted bariatric providers....
Read more detailsDiscover your treatment options with a free, no-obligation quote!
Get your quote now!The Ultimate Medical Tourism Packing Checklist: Preparing for Your Tijuana Hysterectomy
Overview
Patients traveling for a hysterectomy in Mexico should prepare carefully by organizing passports, medical records, prescription medications, loose clothing, hygiene supplies, comfort items, and post-operative travel support. Tijuana is highlighted as a convenient medical tourism hub near the U.S. border, where patients can explore lower-cost hysterectomy options while receiving consultation-based care. The article stresses that patients should confirm treatment suitability, understand recovery needs, and consult qualified healthcare providers before making medical decisions....
Read more detailsTop 5 Reasons Why Americans Choose Tijuana for Advanced Weight Loss Surgery
Overview
Weight loss surgery in Tijuana, Mexico, offers internationally accredited bariatric procedures like gastric sleeve and bypass, combining advanced surgical care, affordable pricing, and streamlined logistics for American patients seeking safe, effective weight management solutions....
Read more detailsKnee Replacement in Tijuana: The Ultimate Guide to Cost and Packages
Overview
Knee replacement surgery in Tijuana, Mexico, offers uninsured and underinsured North American patients a safe, high-quality alternative to exorbitant domestic healthcare, typically providing cost savings of 50% to 70%. While a total knee replacement in the US can cost up to $50,000, accredited top-tier border clinics in Tijuana offer the same procedure for $8,000 to $12,000 through transparent, all-inclusive packages that eliminate hidden hospital fees. These comprehensive bundles cover the surgeon, facility, anesthesia, and premium FDA-approved prosthetics from global leaders like Zimmer Biomet and Stryker. By combining board-certified surgical expertise, rigorous international safety protocols, and dedicated post-operative care, Tijuana provides accessible joint restoration that allows patients to reclaim their mobility without facing overwhelming medical debt....
Read more details4 Top Destinations with Trusted Dental Veneer Clinics in Colombia
Overview
Colombia has become a prime destination for patients seeking premium porcelain veneers, combining skilled cosmetic dentists, advanced technology, and affordable pricing. Leading cities include Bogota, Medellin, Cali, and Cartagena, each offering high-quality clinics that cater to international patients. With comprehensive smile makeover services, short treatment durations, and significant savings compared to North America, Colombia provides both aesthetic excellence and an attractive medical tourism experience....
Read more detailsDiscover your treatment options with a free, no-obligation quote!
Get your quote now!Top 5 Reasons International Patients Travel to Dubai for Family Balancing
Overview
Dubai has become a preferred destination for family balancing thanks to its advanced fertility technologies, internationally experienced specialists, and patient-focused care. Many international patients travel to Dubai to access IVF combined with preimplantation genetic testing (PGT), allowing for embryo selection as part of a broader family planning strategy. Along with modern clinics, high medical standards, and convenient global connectivity, Dubai offers a comprehensive experience for families exploring reproductive treatment options....
Read more detailsThe Complete Guide to Medical Tourism for Fertility Treatments in the UAE
Overview
The United Arab Emirates has emerged as a leading destination for fertility medical tourism, attracting patients with advanced reproductive technologies, internationally trained specialists, and modern healthcare facilities. From IVF and ICSI to fertility preservation and genetic testing, the UAE offers a wide range of treatment options for couples and individuals seeking reproductive care. Understanding treatment costs, success rates, legal requirements, and clinic selection can help international patients make informed decisions and plan a smoother fertility journey....
Read more details5 Things International Patients Don't Know About Plastic Surgery in Thailand
Overview
Thailand has become one of the world’s most established destinations for plastic surgery, attracting patients from across the globe. While affordability is often the first attraction, many visitors are surprised by the level of medical expertise, internationally accredited facilities, personalized patient care, and comprehensive recovery services available. Understanding these lesser-known aspects can help international patients make more informed decisions when considering cosmetic surgery in Thailand....
Read more details5 Surprising Things International Patients Should Know About Puerto Vallarta’s Medical Tourism Industry
Overview
Puerto Vallarta has quietly become one of Mexico’s fastest-growing medical tourism destinations. While many international patients are initially drawn by lower treatment costs, they often discover modern healthcare facilities, experienced physicians, shorter wait times, and a recovery environment that combines quality care with a world-renowned coastal setting. These factors are helping Puerto Vallarta earn a growing reputation among patients from the United States and Canada seeking treatment abroad....
Read more detailsTop 5 Surprising Realities of Modern Joint and Spine Surgery in Mexico
Overview
Mexico has emerged as a trusted destination for joint and spine surgery, offering patients access to advanced orthopedic care at a fraction of the cost often seen elsewhere. Beyond affordability, many hospitals feature modern surgical technologies, internationally trained specialists, and streamlined treatment pathways that reduce waiting times. These advantages are helping reshape perceptions of orthopedic care in Mexico and attracting a growing number of international patients seeking quality outcomes and personalized care....
Read more detailsK-Beauty Beyond Skincare: The Rise of South Korea’s Plastic Surgery Industry
Overview
Seoul has transformed from a K-beauty trendsetter into one of the world’s most influential plastic surgery destinations. Driven by advanced cosmetic technology, highly specialized surgeons, and a strong culture of aesthetic innovation, South Korea continues to attract patients from across the globe. From facial contouring and skin rejuvenation to minimally invasive procedures, the country’s beauty industry now represents a powerful combination of medical expertise, wellness, and modern cosmetic trends....
Read more detailsHow Mexico Is Winning the North American Plastic Surgery Market
Overview
Mexico has rapidly emerged as a major player in the North American plastic surgery market, attracting thousands of patients from the United States and Canada each year. Competitive pricing, internationally trained surgeons, modern medical facilities, and shorter wait times have positioned Mexico as a top destination for cosmetic procedures. As medical tourism continues to grow, cities like Tijuana, Cancún, and Mexico City are becoming global hubs for affordable and high-quality plastic surgery treatments....
Read more detailsWhy Elite Medical Tourists Are Skipping Beverly Hills for Mexico City
Overview
Some high-end medical travelers are starting to look beyond Beverly Hills and compare Mexico City for cosmetic procedures, recovery experiences, and private medical care. For many patients, the appeal is not only lower pricing. They are also looking at surgeon access, discreet recovery settings, shorter scheduling times, and luxury hospitality during treatment. Mexico City has become part of that conversation because some clinics now market VIP-style experiences that combine surgery, private nursing, wellness services, recovery suites, and concierge travel support for international patients. Still, luxury branding alone does not guarantee quality care. Patients should compare surgeon credentials, hospital accreditation, safety protocols, anesthesia standards, and post-operative support before making a decision abroad....
Read more detailsBeyond the Scalpel: 5 Surprising Realities of Stem Cell Therapy in Thailand
Overview
Many international patients look into stem cell therapy in Thailand hoping to avoid surgery or explore regenerative treatment options for orthopedic injuries, chronic pain, wellness support, or recovery-related conditions. What often surprises them is how much the experience depends on evaluation, treatment planning, and follow-up care not just the procedure itself. Some clinics focus heavily on patient comfort, recovery programs, rehabilitation support, and wellness-based care alongside stem cell therapy. Others attract patients because of shorter waiting times, private hospital settings, or medical tourism services designed for international travelers. At the same time, treatment methods, cell sources, safety standards, and medical oversight can vary between clinics. Patients should ask detailed questions about how cells are prepared, what conditions are evaluated, what results are realistically expected, and what follow-up support is included before traveling to Thailand for treatment....
Read more detailsBeyond the Conventional: 7 Surprising Truths About Cancer Care in Tijuana, Mexico
Overview
For many international patients, cancer care in Tijuana is no longer viewed only as an alternative option. Some patients travel there looking for integrative treatment plans, shorter waiting times, personalized support, or therapies that may not be easily accessible or affordable in their home country. What surprises many people is how broad the care landscape has become. Some clinics combine conventional oncology with nutrition programs, immune-based therapies, wellness support, hyperthermia, or recovery-focused care. Others focus heavily on quality of life, patient comfort, and long-term support during treatment. Patients considering cancer care in Tijuana should still research carefully. Treatment approaches, medical oversight, therapy standards, and clinic experience can vary widely. Asking detailed questions about diagnosis review, treatment goals, safety monitoring, follow-up care, and realistic expectations can help patients make more informed decisions before traveling abroad....
Read more details7 Crucial Questions to Ask Before Choosing an Ibogaine Clinic Abroad
Overview
Choosing an ibogaine clinic abroad is a serious medical and personal decision. Many patients compare programs in places like Mexico because they are looking for addiction treatment options that feel more accessible, private, or specialized. But not every clinic offers the same level of medical care or safety support. Before booking treatment, patients should ask about heart screening, emergency response protocols, medical supervision during detox, staff experience, psychological support, aftercare planning, and what happens if complications occur during treatment. It is also important to understand who is actually overseeing the program and whether the clinic evaluates patients individually before accepting them. The best decision usually comes from asking detailed questions, understanding the risks clearly, and choosing a clinic that prioritizes patient safety and long-term recovery not just fast treatment access....
Read more detailsTable of Contents
- Endometriosis Treatment Abroad: Why Patients Choose Tijuana for Specialized Care
- Regaining Weight After Surgery? 5 Signs You Need a Gastric Sleeve Revision in Tijuana, Mexico
- Escaping US Medical Debt: Why Hernia Surgery in Mexico is the Best Option
- Budgeting For Your Medical Trip: Hidden Savings in Tijuana Hip Surgery
- Comparing Gastric Bypass Costs: Out-of-Pocket Prices in US Hospitals vs Tijuana Mexico












.jpg)





.jpg)

.jpg)



.jpg)
.png)
.png)
.png)

.png)
.png)
.png)
.png)
.png)
.png)








