内容
重塑长期康复:日本的再生医学
通过先进的医学技术恢复活力、对抗退行性疾病已成为众多国际患者的现实。在日本,干细胞疗法已成为寻求精准、细致地激发人体自身修复潜能的患者的首选之地。日本以其严格的安全标准和临床创新而闻名,将卓越的传统与先进的医疗研究基础设施相结合,使其成为全球患者的首选干细胞疗法。
日本汇聚了一批专注于再生医学的顶尖研究人员和临床医生。这些从业者往往是该领域的先驱,并在全球最先进的监管框架下开展工作。在干细胞领域,日本脱颖而出,因为它提供高端医疗体验,拥有利用诱导多能干细胞 (iPSCs) 和其他突破性先进细胞疗法技术的设施。
在日本,再生疗法不仅仅是医疗干预,更在于通过细胞修复来恢复生活质量。无论您是希望治疗骨科损伤、神经系统疾病,还是抗衰老,这里的专家都将安全性和长期疗效放在首位。选择这个科技发达的国家,患者不仅可以接受变革性的治疗,还能体验到世界最具创新精神的社会之一的独特文化和热情好客。
你可知道?
日本已转型成为全球医疗创新中心,尤其是在细胞领域。山中伸弥博士凭借诱导多能干细胞(iPS细胞)的发现荣获诺贝尔奖,加速了这一发展进程,并促使日本对再生医学进行了大量投资。这确保了患者能够获得目前全球市场上监管最严格、科学验证最充分的再生疗法。
关键信息概览
由于日本的干细胞疗法受《再生医学安全法》(ASRM)的监管,因此可以期待最高的安全性。
日本顶尖医生引领着干细胞研究,确保为复杂的慢性疾病提供全面的专业知识。
这里的专家使用专有的细胞扩增方法,非常适合实现高效的日本再生医学效果。
日本对先进细胞疗法制定了严格的伦理准则,确保患者获得合法且有科学依据的治疗。
顶尖医疗团队将配备专门的协调员,全程支持您在日本的再生疗法之旅。
患者受益于注重精准和洁净的环境,营造出宁静的氛围,有助于在日本干细胞治疗后恢复健康。
精准文化
当您专注于自身健康时,您身处一个以精准为生活方式的国度。从举世闻名的新干线列车的准点率,到实验室一丝不苟的严谨操作,日本对卓越的不懈追求为您的医疗之旅营造了独特的氛围,带来可靠的保障,从而真正助力您的康复。
为了简化您的就医流程,东京和大阪的许多顶级诊所都提供专为国际患者设计的综合套餐。这些套餐通常包括医疗咨询、实验室检测、在专业设施中进行细胞处理以及治疗实施。以下我们整理了一些信息,帮助您了解选择日本这个创新国家进行干细胞治疗的优势,以及您在日本接受再生医学治疗的具体目标。
注意:在日本预约再生疗法时,许多患者选择采用日本先进细胞疗法方案,以确保获得最高质量的日本干细胞,从而获得康复和长期健康效果。
了解在日本进行干细胞治疗的投资,需要参考日本再生医学的平均价格,通常根据治疗病症的不同,价格在 12,000 美元到 25,000 美元之间。考虑到日本高水平的监管和安全保障,这一价格具有竞争力,尤其与其他提供先进细胞疗法的国家相比更是如此。这些费用涵盖了在日本获得许可的细胞处理中心进行干细胞培养所需的复杂流程。您在日本接受再生疗法的最终价格将取决于细胞来源、所需的注射次数以及为您制定的具体临床方案。您可以利用这些信息,自信地规划您的专业医疗预算。
提示:在计算总预算时,请记住将初始诊断成像费用和 5-7 天的细胞培养期费用考虑在内。
Stem Cell Therapy 中心成本比较 Japan
| 提供者 | 程序 | 价格 |
|---|---|---|
| HELENE - Stem Cell Clinic | Stem Cell Treatment for Diabetes, Stem Cell Therapy | $10000 |
Stem Cell Therapy Cost Comparison in Japan
| 国家 | 程序 | 价格 |
|---|---|---|
| United States | Stem Cell Treatment for Diabetes, Stem Cell Therapy | $22000 |
日本凭借其众多资质认证的研究人员和一流的医疗基础设施,已成为干细胞治疗的首选目的地。在日本寻求干细胞治疗的患者可以选择高端医疗中心,这些中心提供间充质干细胞治疗和再生医学等先进技术。这些医疗机构旨在为国际患者提供无缝衔接的就医体验,将卓越的医疗水平与日本先进细胞治疗所特有的精准服务和热情周到的关怀完美融合。
除了治疗本身,选择在日本接受再生疗法还能让患者获得世界一流的医疗服务,走在生物创新的前沿。在计划前往日本进行干细胞治疗时,务必调查医疗团队的资质和医疗机构的认证情况,以确保在日本干细胞治疗过程中获得最高的安全性和疗效。
安全第一:核实该诊所是否已获得日本厚生劳动省根据《再生医学安全法》颁发的许可证,以确保其符合严格的国家安全和道德标准。
亲眼见证治疗效果,能让您在日本考虑干细胞疗法时更加清晰明了,信心倍增。本部分精选了一系列患者视频,展示了真实的治疗效果和个人历程。这些视频清晰地呈现了日本再生医学的全过程,从您抵达诊所到最终康复,全程记录。通过观看这些患者证言,您可以更深入地了解日本先进细胞疗法的卓越品质,以及日本干细胞和再生疗法专家提供的个性化护理。
建议:在观看有关日本再生医学的视频时,请密切关注患者提到的长期康复里程碑,以帮助您对自己在日本接受干细胞治疗后的康复时间表设定切合实际的期望。
下方列出了曾在我们合作诊所接受过日本干细胞治疗的患者的真实评价和评分。这些评价深入剖析了日本再生医学的疗效、医护人员的专业水平以及前往日本接受干细胞治疗的整体体验。阅读这些患者证词,可以帮助您更清晰地了解在日本接受先进细胞疗法和再生疗法的预期效果,从而帮助您选择合适的日本干细胞治疗专家。
审核提示:请留意有关初次咨询的深度以及诊所在使用的具体细胞类型和预期功能改善时间表方面的透明度。
日本的干细胞疗法安全吗?
是的,由于日本严格的监管体系,干细胞疗法被认为是世界上最安全的疗法之一。《再生医学安全法》确保所有提供再生医学服务的日本诊所都遵守严格的细胞处理和患者护理标准。选择日本先进细胞疗法的患者可以信赖一个将临床安全和伦理透明度放在首位的体系。
日本干细胞疗法通常治疗哪些疾病?
日本的医疗机构利用干细胞疗法治疗多种疾病,包括骨关节炎、脊髓损伤和心血管疾病。再生疗法在日本也被用于治疗自身免疫性疾病和抗衰老。日本各高级细胞治疗中心均专注于不同的治疗方案,确保患者获得针对其特定医疗需求的精准治疗。
日本再生医学的独特之处是什么?
日本之所以能成为全球领先者,是因为它是首个专门针对再生医学实施快速审批制度的国家。这使得患者能够比许多其他国家的患者更早地获得先进的细胞疗法。此外,尖端技术的融合和高标准的实验室条件,也使日本的干细胞疗法成为国际患者的首选。
在日本进行干细胞治疗的费用是多少?
费用根据所用细胞类型和病情复杂程度而有所不同,但日本的干细胞疗法凭借其高水平的技术,价格极具竞争力。虽然可能比一些发展中地区更贵,但日本在再生疗法领域的专业技术却能带来显著的价值。在日本接受先进的细胞疗法通常被视为对健康和行动能力的一项长期投资。
在日本接受再生疗法需要多长时间?
大多数在日本接受再生疗法的患者应计划停留5至10天。这段时间足以完成初次咨询、细胞输注以及离境前的必要监测。对于某些需要细胞培养的干细胞疗法,患者可能需要两次就诊,或者安排更长的单次停留时间以完成整个疗程。
日本干细胞治疗中使用的是自体细胞还是异体细胞?
两种类型的干细胞疗法均可提供,但日本干细胞中心通常专注于自体干细胞疗法,即使用患者自身的细胞以最大程度地降低排异风险。这是日本再生医学的关键组成部分,确保了高度的相容性。然而,日本先进细胞疗法中心正在进行的研究也包括针对特定临床应用的高质量异体干细胞疗法。
日本先进细胞疗法术后需要恢复期吗?
与传统手术相比,日本先进细胞疗法的恢复期通常非常短。许多在日本接受干细胞疗法的患者几乎无需休息,通常几天内即可恢复轻度活动。日本大多数再生疗法的非侵入性特点,使得患者能够经历一个渐进且自然的愈合过程,并在几个月内逐渐好转。
我如何知道自己是否符合日本再生医学项目的申请条件?
在日本,再生医学的适用人群通常是寻求非侵入性手术治疗方案的患者,或是患有慢性疾病且对常规治疗无效的患者。患者需接受日本干细胞专家的详细医学评估,以确定其是否符合治疗条件。日本大多数干细胞治疗机构会要求患者提供近期病历和影像资料,以便进行准确的评估。
日本再生疗法的成功率是多少?
在日本,再生疗法的成功率因病症而异,但通常在疼痛管理和功能改善方面效果显著。选择日本先进细胞疗法的患者通常表示症状显著减轻,生活质量得到改善。日本在干细胞疗法领域的临床专长确保患者能够获得目前最有效的治疗方案。
我如何才能在日本预约干细胞治疗?
您可以通过 PlacidWay 平台联系日本的认证诊所,预约干细胞疗法。该平台可帮助您找到日本再生医学领域的顶尖专家,比较不同的治疗方案,并安排咨询。无论您是对日本干细胞疗法还是最新的先进细胞疗法感兴趣,PlacidWay 都为国际患者简化了预约流程。
How Much Does Stem Cell Therapy Cost in Japan?
The cost of stem cell therapy in Japan typically ranges from $10,000 to over $40,000 USD per treatment, depending on the condition, cell type, and clinic. Thinking about exploring advanced medical treatments abroad? Japan has become a global leader in regenerative medicine, particularly stem cell therapy. This reputation for high-tech innovation and strict safety standards makes it a top destination for patients worldwide. But the big question on everyone's mind is: how much does it actually cost? The price for stem cell therapy in Japan isn't a single number; it's a range influenced by many factors, from the specific condition you're treating to the type of stem cells used. This guide is here to break down those costs for you. We'll dive into what you're paying for, what kinds of treatments are available, and why Japan's approach is considered so cutting-edge. Whether you're considering treatment for joint pain, anti-aging, or more complex conditions, understanding the financial side is the first step. Let's explore what you can expect to invest in your health at some of the world's most advanced Japanese stem cell clinics. What is the average cost of stem cell therapy in Japan? The average cost for a single stem cell therapy session in Japan typically falls between $10,000 and $40,000 USD (approximately 1.5 to 6 million JPY). Comprehensive anti-aging or neurological packages can be higher. This price range is a general estimate. For example, a localized treatment, like an injection for knee osteoarthritis, will be on the lower end of that scale. In contrast, a systemic intravenous (IV) treatment for anti-aging, longevity, or autoimmune conditions will be at the higher end. This is because systemic treatments often require a much higher number of cultured cells, and the lab work involved is more extensive. Many clinics in Japan offer packages that might include the initial consultation, blood work, cell harvesting (if using your own cells), cultivation, and the final administration. Always get a detailed quote that outlines exactly what is included to avoid any surprises. What factors influence the final price? Several key factors determine the total cost of stem cell therapy in Japan: the type of stem cells used (e.g., adipose, umbilical), the number of cells administered, the condition being treated, the clinic's reputation, and the number of sessions required. Source of Stem Cells: Cells can be autologous (your own) or allogeneic (from a donor). Autologous cells from your own fat (adipose) or bone marrow require harvesting and a cultivation period (often 4-6 weeks), which adds to the cost. Allogeneic cells, like those from umbilical cord tissue, are often "off-the-shelf" but have their own associated sourcing and screening costs. Cell Count: The number of cells, often counted in the millions (e.g., 100 million vs. 250 million), directly impacts the price. More cells mean more extensive lab cultivation. Treatment Complexity: A simple joint injection is less complex and cheaper than an IV infusion or a multi-site injection protocol. Clinic & Location: Top-tier clinics in major cities like Tokyo or Osaka with advanced research facilities and leading doctors may charge more. Cost Comparison: Stem Cell Therapy in Japan (Estimates) A detailed cost comparison shows that anti-aging and systemic treatments are generally the most expensive, while localized joint treatments are more affordable. Prices can range from 1,000,000 JPY for a single knee to over 6,000,000 JPY for systemic IV therapies. Here is a sample table of estimated costs for common treatments available at Japanese stem cell clinics. These prices are estimates and can vary significantly between clinics. (Note: USD conversions are approximate and subject to currency fluctuation). Treatment Type Condition Treated Estimated Cost (JPY) Estimated Cost (USD) Local Injection (Knee) Osteoarthritis (One Knee) ¥1,000,000 - ¥1,500,000 $6,500 - $9,700 Local Injection (Knees) Osteoarthritis (Both Knees) ¥1,400,000 - ¥2,000,000 $9,000 - $13,000 Local Injection (Joints) Erectile Dysfunction (ED) ¥2,200,000 - ¥2,800,000 $14,200 - $18,000 Aesthetic / Skin Facial Rejuvenation (Skin) ¥1,000,000 - ¥1,600,000 $6,500 - $10,300 Systemic IV Infusion Anti-Aging / Longevity ¥3,400,000 - ¥6,000,000+ $22,000 - $38,800+ Systemic IV Infusion Motor Function / Neurological ¥1,300,000 - ¥4,500,000 $8,400 - $29,000 Systemic IV Infusion (Package) Autoimmune / Degenerative ¥4,400,000 - ¥9,900,000+ $28,400 - $64,000+ Why is stem cell therapy in Japan considered so advanced? Japan's reputation in regenerative medicine is built on its pioneering research, especially in Induced Pluripotent Stem Cells (iPS cells), and its strict regulatory framework. The government fast-tracks promising therapies, fostering rapid innovation in a safe, controlled environment. In 2014, Japan's government revised its laws to create a unique, fast-track approval system for regenerative medicine. This allows clinics to offer therapies to patients on a conditional basis after safety and efficacy have been shown, rather than waiting for decades of large-scale trials. This environment, combined with Japan’s pioneering research on induced pluripotent stem cells (iPS cells), has positioned the country’s stem cell clinics at the forefront of medical science. These clinics operate with advanced laboratory technology and under strict oversight from the Ministry of Health, Labour and Welfare (MHLW). Is stem cell therapy legal and safe in Japan? Yes, stem cell therapy in Japan is legal and operates under one of the world's most comprehensive regulatory frameworks. The "Act on the Safety of Regenerative Medicine" ensures that clinics meet stringent standards for cell processing, administration, and patient safety. This law requires all clinics to submit their treatment plans to the MHLW for approval. This means the government is aware of what treatments are being offered and monitors them for safety. This is a key reason why international patients trust Japan for these advanced procedures. While all medical procedures carry some risk (like infection or treatment failure), Japan's strict oversight minimizes these risks significantly compared to countries with little to no regulation. What types of stem cells are used in Japanese clinics? The most common types used in Japanese stem cell clinics are Mesenchymal Stem Cells (MSCs). These are sourced from the patient's own adipose (fat) tissue or bone marrow, or from a donor's umbilical cord tissue. Adipose-Derived Stem Cells (ADSCs): These are autologous (from your own body). A doctor performs a mini-liposuction to collect a small amount of fat. These cells are then isolated and cultured for several weeks to multiply into the millions before being reinjected. Bone Marrow-Derived Stem Cells (BMDSCs): Also autologous, these are collected from the hip bone. This method is slightly more invasive than fat collection. Umbilical Cord-Derived Stem Cells (UCSCs): These are allogeneic (from a donor) and are collected from the tissue of donated umbilical cords after a healthy birth. They are highly potent and versatile, and using them avoids a harvesting procedure for the patient. What is the stem cell therapy process like in Japan? The typical process involves an initial consultation, pre-treatment testing, cell harvesting (if autologous), cell cultivation in a lab, and finally, the administration of the cells via injection or IV infusion, followed by a brief recovery and follow-up. Here’s a step-by-step look: Consultation: You'll speak with a specialist (often remotely at first) to discuss your medical history and goals. Pre-Treatment: Once in Japan, you'll undergo blood tests and a physical exam to ensure you're a good candidate. Harvesting (if needed): If using your own cells, a doctor will collect fat or bone marrow. This is a minor outpatient procedure. Cultivation: This is the waiting period. Your cells are sent to a specialized lab (a "Cell Processing Center") where they are grown for 4-6 weeks. Many international patients return home during this time. Administration: You'll return to the clinic to receive your cultured cells. This is usually a simple procedure (a joint injection or an IV drip) that takes 30-60 minutes. Follow-up: You'll be monitored for a short period before being cleared to leave. The clinic will typically schedule follow-up calls or visits. Does Japanese health insurance cover stem cell therapy? Generally, no. For most conditions like anti-aging, aesthetic treatments, and osteoarthritis, stem cell therapy in Japan is considered an elective, out-of-pocket expense and is not covered by Japanese national health insurance or most international insurance plans. While Japan has approved specific stem cell-based products for certain critical diseases (like stroke or graft-versus-host disease) which may be covered by their national insurance, the treatments sought by medical tourists fall outside of this. You should always assume you will be paying for the procedure yourself. What conditions are treated with stem cell therapy in Japan? Japanese stem cell clinics treat a wide range of conditions, with the most common being osteoarthritis (especially knees and hips), anti-aging and longevity, aesthetic concerns (skin rejuvenation and hair loss), sports injuries, and some autoimmune and degenerative conditions. The anti-inflammatory and regenerative properties of stem cells are what make them so versatile. For joints, they can help reduce pain and inflammation, potentially repairing cartilage. For anti-aging, systemic IV infusions are believed to reduce inflammation throughout the body, improve energy levels, and enhance overall wellness. Research is also active for more complex conditions like Parkinson's, stroke recovery, and diabetes. What are stem cell 'supernatants' or 'exosomes'? Exosomes, or the 'culture supernatant,' are byproducts of stem cell cultivation. They are not cells, but tiny vesicles that contain the growth factors, proteins, and signaling molecules that stem cells use to communicate. These are often offered as a separate, more affordable acellular (cell-free) therapy. Think of it this way: if stem cells are the "factory," exosomes are the "packages" they send out that tell other cells what to do (e.g., "reduce inflammation" or "build new tissue"). These treatments are popular for aesthetics and skin quality, as they provide many of the regenerative signals without the cells themselves. This can be a good, lower-cost alternative for some patients. How long do I need to stay in Japan for treatment? It depends on the treatment. If you use donor (allogeneic) cells, you may only need to be in Japan for 3-7 days for the consultation and treatment. If you use your own (autologous) cells, you will need to make two trips: one short trip for harvesting, and a second trip 4-6 weeks later for the administration. Many patients prefer the two-trip protocol. The first trip is just 2-3 days for the consultation and fat harvesting. They then fly home and return for another 2-3 day trip for the injection or infusion. This breaks up the travel and is very efficient. Clinics are very experienced with international patients and can help coordinate this schedule. How do I choose a good stem cell clinic in Japan? Look for clinics that are officially registered with the Japanese Ministry of Health, Labour and Welfare (MHLW). A reputable clinic will be transparent about its registration, the source and type of its cells, and its pricing. Patient reviews and a direct consultation are also crucial. Here's what to check for: MHLW Approval: Ask the clinic for its "Notification Number" from the MHLW. This is proof they are legally permitted to perform regenerative medicine. Experienced Staff: Check the credentials of the doctors and specialists. In-House or Certified Lab: Do they have their own high-grade "Cell Processing Center" (CPC) or use a certified partner? This is critical for cell quality. Transparency: They should provide a clear, itemized quote and patiently answer all your questions about the procedure, risks, and realistic outcomes. What are the risks or side effects? Side effects from stem cell therapy in Japan are generally minimal and rare, especially when performed at a certified clinic. The most common are temporary pain, swelling, or bruising at the injection site. With IV infusions, some patients report mild, flu-like symptoms or fatigue for a day. Because Japan's regulations are so strict, the risk of serious complications like infection or contamination is extremely low. If using autologous cells (your own), the risk of rejection or allergic reaction is virtually zero. Using donor cells (allogeneic) carries a theoretical, but very low, risk of reaction, which is why all donor cells are rigorously screened. How soon will I see results? This varies by patient and condition. Some patients report feeling reduced pain and inflammation within a few weeks. However, because stem cells work by regenerating tissue, the full, significant results often develop over 3 to 6 months as the cells work to repair and rebuild the damaged area. Regenerative medicine is not an instant fix. It's a biological process. For joint pain, you might feel initial relief quickly, but the more substantial, lasting improvement in mobility and pain reduction builds over several months. For anti-aging or systemic treatments, benefits like improved energy, better sleep, and enhanced skin quality are also typically reported gradually over months. Is Japan cheaper than the US for stem cell therapy? Yes, often significantly. While stem cell therapy is available in the US, the FDA has not approved it for most conditions, so treatments are limited and can be extremely expensive, often $20,000 to $50,000 or more for procedures that are still considered experimental. Japan's regulatory environment allows for these therapies to be offered more widely and, in many cases, at a more competitive price point. Patients travel to Japan not just for the lower cost, but because they are getting access to highly advanced, government-regulated therapies that simply aren't available in their home country. Ready to explore your options for high-quality, advanced healthcare? Don't let uncertainty hold you back. PlacidWay connects you with a network of world-class, accredited medical facilities in Japan and around the globe.
Is Stem Cell Therapy Legal in Japan?
Yes, stem cell therapy is legal in Japan. It is regulated under a specific national law, the "Act on the Safety of Regenerative Medicine" (ASRM), which was passed in 2014. When you hear about cutting-edge medical treatments, "stem cell therapy" and "Japan" often come up in the same sentence. Japan has positioned itself as a world leader in regenerative medicine, attracting patients from all over the globe. But with so much conflicting information online, one of the first and most important questions people ask is: "Is stem cell therapy even legal in Japan?" The simple answer is a resounding yes. Not only is it legal, but it's also regulated by a comprehensive, forward-thinking legal framework that sets it apart from almost every other country. This isn't a medical "wild west." The Japanese government has created specific laws to safely fast-track promising therapies. This means that treatments that might be considered "experimental" and only available in restrictive clinical trials in the US or Europe can be legally offered in Japanese stem cell clinics. This guide will walk you through exactly what makes it legal, what the regulations are, and what you need to know to safely navigate your options for stem cell therapy in Japan. What makes stem cell therapy legal in Japan? Stem cell therapy is legal in Japan under the "Act on the Safety of Regenerative Medicine" (ASRM), which was passed in 2014. This law creates a specific regulatory framework for offering these treatments outside of conventional, long-term clinical trials. This law is what makes Japan unique. Instead of the long, multi-decade approval process required by agencies like the US Food and Drug Administration (FDA), Japan's ASRM creates a different pathway. It allows clinics to offer regenerative therapies to patients after their treatment plan has been reviewed for safety and rationale by an independent, government-accredited committee. This system is designed to accelerate the availability of safe, promising treatments. The trade-off is that clinics must meticulously track patient data and safety outcomes and report them to the Ministry of Health, Labour and Welfare (MHLW). This allows the government to gather real-world evidence on these therapies while giving patients access to them sooner. What is the "Act on the Safety of Regenerative Medicine" (ASRM)? The ASRM is a Japanese law enacted in 2014 that allows clinics to provide stem cell therapies after a government-accredited committee reviews their treatment plan for safety and efficacy, creating a fast-track, regulated pathway. This law classifies regenerative medicine treatments into three risk categories (Class I, II, and III). Most treatments offered to medical tourists, like using a patient's own fat-derived stem cells, fall into the lower-risk categories (Class II or III). To offer any of these treatments, a clinic can't just open its doors and start injecting. They must first submit a detailed "treatment plan" to an independent review board. This plan outlines: What kind of cells they will use (e.g., adipose-derived, umbilical cord-derived). How the cells will be processed and grown. What condition they are treating (e.g., knee osteoarthritis). All the safety protocols involved. Only after this committee approves the plan and it's registered with the MHLW can the clinic legally offer that specific therapy. Is stem cell therapy in Japan safe and regulated? Yes, stem cell therapy in Japan is highly regulated for safety. The MHLW requires all clinics to report on their procedures, use certified cell processing centers, and track patient outcomes, ensuring a high standard of care. The legality of the therapy is directly tied to its safety. The ASRM mandates that all cell processing (the separating, growing, and storing of stem cells) must be done in a Cell Processing Center (CPC). A CPC is a highly specialized, clean-room laboratory that must meet stringent government standards for quality control, purity, and safety. This is a crucial point. It prevents clinics from processing cells in an unsterile back room, which is a major risk in unregulated markets. This focus on a sterile, high-quality "product" is a cornerstone of Japan's safety regulations and a key reason why patients trust the system. Why is Japan a leader in stem cell research? Japan is a leader in regenerative medicine thanks to strong government support, major investment in research, and the groundbreaking 2012 Nobel Prize-winning discovery of induced pluripotent stem cells (iPS cells). The 2012 Nobel Prize in Medicine marked a turning point for Japan, sparking national enthusiasm and pride in regenerative medicine. This milestone led to significant government and private investment in the field. The resulting research momentum ultimately inspired the creation of the ASRM, designed to safely and efficiently translate scientific breakthroughs into real-world patient treatments. What are iPS cells (induced pluripotent stem cells)? iPS cells are a type of stem cell "created" in a lab, typically from a patient's own skin or blood cells. They are "reprogrammed" back into a blank-slate state, allowing them to become any other cell type in the body. Think of it this way: an adult skin cell is "specialized" and can only be a skin cell. Dr. Yamanaka discovered how to hit a "factory reset" button on that cell, turning it back into a "pluripotent" stem cell—one that is just like an embryonic stem cell and has the potential to become a heart cell, brain cell, or cartilage cell. This discovery is the future, but it's important to know that most legal therapies offered to patients today use Mesenchymal Stem Cells (MSCs), not iPS cells. What's the difference between Japanese and US stem cell regulations? The main difference is the pathway to approval. Japan has a conditional, fast-track approval system (ASRM) that allows patients to access therapies sooner. In the US, the FDA generally requires long, large-scale clinical trials before a therapy is approved for wide use. In the United States, most stem cell therapies (outside of a few specific bone marrow and cord blood uses) are classified as "investigational new drugs." This means they must go through the same rigorous, multi-phase clinical trial process as any new pharmaceutical, which can take 10-15 years and hundreds of millions of dollars. Because of this, most stem cell treatments in the US are only legally available to patients who are enrolled in a clinical trial. Japan's system, in contrast, says that if a therapy has shown a strong safety profile and has a logical scientific basis, it can be offered to paying patients *while* the clinic gathers more data. This is a fundamental shift in philosophy, prioritizing patient access alongside safety and data collection. How do I know if a Japanese stem cell clinic is legitimate? A legitimate clinic in Japan will be registered with the Ministry of Health, Labour and Welfare (MHLW) and have an official "Notification Number" for their specific treatment plan. Always ask for this proof of registration. Here are a few things to check to verify a clinic is legal and reputable: Ask for their MHLW Notification Number: This is non-negotiable. A legal clinic will have this and should be transparent about it. Confirm they use a certified CPC: Ask them where your cells will be processed. A legitimate clinic will proudly name their certified lab partner. Look for transparency: They should provide a clear, itemized quote and patiently answer all your questions about risks and realistic outcomes. Beware of "Miracle Cures": Reputable doctors are scientific and cautious. Be wary of any clinic that guarantees 100% success or claims to cure everything. What types of stem cell therapies are legal and common in Japan? The most common legal therapies in Japan use Mesenchymal Stem Cells (MSCs). These are typically sourced from the patient's own adipose (fat) tissue, bone marrow, or from a donor's umbilical cord tissue. These MSCs are "adult" stem cells, which are non-controversial and have a strong safety profile. They are known for their powerful anti-inflammatory and regenerative signaling abilities. They work by quieting inflammation and signaling your body's own repair mechanisms to get to work. What conditions do Japanese clinics legally treat with stem cells? Japanese clinics legally treat a wide range of conditions, most commonly osteoarthritis (especially knees), anti-aging and wellness, sports injuries, skin rejuvenation, and are researching treatments for autoimmune and neurological conditions. Because the law is flexible, clinics can submit treatment plans for many different conditions. The most common and well-established treatments you will find are for orthopedic issues like knee, hip, or shoulder pain. Systemic IV infusions for general wellness, anti-aging, and to support autoimmune conditions are also very common. How much does legal stem cell therapy cost in Japan? The cost of legal stem cell therapy in Japan varies widely, from around $6,500 for a single joint injection to over $40,000 for comprehensive anti-aging or neurological IV treatments. The price depends on the cell type and quantity. The high cost is a direct reflection of the strict legal and safety standards. The price includes the doctor's expertise, the minor harvesting procedure (if needed), and, most importantly, the expensive work done at the certified Cell Processing Center (CPC) to isolate, culture, and test your cells for purity and viability. Cost Comparison of Legal Stem Cell Treatments in Japan (Estimates) Prices for legal treatments range from approximately 1,000,000 JPY for localized injections to over 6,000,000 JPY for systemic IV infusions, reflecting the high safety and lab standards required by Japanese law. Treatment Type Condition Treated Estimated Cost (JPY) Estimated Cost (USD) Local Injection (Adipose MSCs) Osteoarthritis (One Knee) ¥1,000,000 - ¥1,500,000 $6,500 - $9,700 Systemic IV Infusion (Adipose MSCs) Anti-Aging / Wellness ¥3,400,000 - ¥6,000,000 $22,000 - $38,800 Local Injection (Fibroblasts) Skin Rejuvenation (Face) ¥1,000,000 - ¥1,600,000 $6,500 - $10,300 Systemic IV Infusion (Umbilical MSCs) Autoimmune Support ¥4,400,000 - ¥9,900,000+ $28,400 - $64,000+ Is it safe for a foreigner to get stem cell therapy in Japan? Yes, it is generally very safe for foreigners. Reputable Japanese clinics are accustomed to international patients and must adhere to the same strict MHLW safety regulations for all patients, regardless of origin. The legal framework protects everyone, not just Japanese citizens. Top-tier clinics often have dedicated international patient coordinators and English-speaking staff to ensure a smooth and safe experience. You are protected by the same laws and quality controls as a local patient. What are the risks of stem cell therapy, even if it's legal? Even though it's legal and regulated, minimal risks exist. These are typically minor, such as temporary pain, swelling, or bruising at the injection or harvesting site. The risk of serious complications like infection is extremely low due to Japan's strict lab standards. A reputable doctor will discuss these with you. With autologous (your own) cells, there is no risk of rejection. With allogeneic (donor) cells, they are carefully screened, but a very small risk of allergic reaction could exist. The most significant risk in the global stem cell market—contamination—is the very thing Japan's CPC system is designed to prevent. Does "legal" mean "guaranteed to work"? No. "Legal" means the therapy is permitted and regulated for safety. It does not mean it is a guaranteed cure. Efficacy varies from patient to patient, and reputable clinics will provide realistic expectations. This is a critical distinction. The Japanese system allows these treatments to be offered because they have a strong scientific basis and a high safety profile. However, medicine is complex. A 30-year-old athlete with a minor cartilage tear will likely have a different result than an 80-year-old with severe, bone-on-bone arthritis. A good clinic will assess your specific case and be honest about the potential outcomes. Can I get iPS cell therapy in Japan? Generally, no. While Japan leads iPS cell research, these therapies are almost exclusively in the clinical trial phase for very specific, serious conditions (like macular degeneration or Parkinson's) and are not yet available as a commercial treatment in clinics. This is a common point of confusion. The legal treatments you can pay for as a patient at a private clinic almost always use Mesenchymal Stem Cells (MSCs). The revolutionary iPS cell therapies are still largely in the research and trial stage at major university hospitals and are not yet available to the public. Ready to explore the safe, legal, and advanced options for Do stem cell therapy in Japan? PlacidWay connects you with a network of world-class, accredited medical facilities in Japan and around the globe.
Can Stem Cells Help Recover from Paralysis in Japan?
Yes, recent breakthroughs in Japan show that stem cell therapy, particularly using iPS-derived cells, has helped some patients with spinal cord injuries regain motor function, offering new hope for paralysis recovery. Hello, and welcome! If you or a loved one are facing the challenges of paralysis, you've likely heard whispers of groundbreaking medical advancements. One of the brightest beacons of hope is coming from Japan, a country at the forefront of stem cell therapy. The big question on everyone's mind is, "Does it actually work?" The short answer is that it's showing incredible promise. We're not talking about a far-off science fiction concept anymore; we're talking about real clinical studies where patients with severe spinal cord injuries have regained movement. It's a journey, not a magic wand, but the progress is undeniable and exciting. Japan has uniquely positioned itself as a global leader in this field, thanks to supportive government regulations and world-class research institutions. This has created an environment where cutting-edge treatments can be developed and, in some cases, offered to patients sooner than in other parts of the world. This doesn't mean it's a free-for-all; the process is still carefully regulated, but the focus is on accelerating hope and healing. In this post, we're going to dive deep into what's really happening with stem cell therapy for paralysis in Japan. We'll cut through the hype and look at the facts. What types of treatments are available? What do the studies *actually* say about success rates? Who is a good candidate? And, of course, what is the cost of stem cell therapy? We'll answer the questions you've been searching for, so you can get a clearer picture of what's possible. What is stem cell therapy for paralysis? Stem cell therapy for paralysis is a regenerative treatment that uses special cells (stem cells) to help repair damaged nerve tissue in the spinal cord, reduce inflammation, and potentially restore lost motor or sensory function. At its core, stem cell therapy for paralysis is a form of regenerative medicine. When a spinal cord injury (SCI) occurs, the vital connection between the brain and the rest of the body is damaged or severed. This blocks signals, leading to paralysis. Traditional treatments focus on stabilization and rehabilitation, but they can't repair the damaged spinal cord itself. This is where stem cells come in. Stem cells are like the body's master builders. They have the amazing ability to develop into different types of cells and can also self-renew. When used to treat paralysis, the idea is that these cells can be introduced to the injury site to: Replace damaged nerve cells: Some stem cells can be coaxed into becoming new neurons or the support cells (glia) that help neurons function. Reduce inflammation: Inflammation at the injury site can cause further damage. Many stem cells, particularly Mesenchymal Stem Cells (MSCs), are powerfully anti-inflammatory. Release protective factors: Stem cells secrete special proteins (growth factors) that can protect the surviving nerve cells from dying and encourage them to grow. Modulate the immune system: They can help calm the body's immune response, which sometimes attacks its own tissue after an injury. The goal isn't just to patch a hole. It's to create a more supportive environment for healing, to rebuild broken circuits, and to give the body a chance to repair itself in ways it couldn't on its own. Can stem cells really help paralysis recovery? Yes, clinical studies, particularly in Japan, have shown that stem cells can help some patients with paralysis recovery. In a recent trial, patients with spinal cord injuries regained the ability to stand or use their arms. This is the most important question, and the answer is a cautious but optimistic "yes." For a long time, spinal cord injuries were considered permanent. However, recent breakthroughs are challenging that grim prognosis. The most compelling evidence comes from a clinical trial at Keio University in Tokyo. In this landmark study, researchers treated patients with "subacute" spinal cord injuries (meaning their injury was recent, typically within a few weeks). They injected millions of special stem cells, called iPS-derived neural stem cells, directly into the injury site. The results, published in 2022, were remarkable: out of four patients, two showed significant improvement. One patient, who was fully paralyzed, regained the ability to stand and even practice walking. Another regained the ability to move their arms and feed themselves. This is considered a massive leap forward. It's important to be realistic. This was a small, early-phase study focused primarily on safety. The success rate was "50%" in this tiny group, and the other two patients did not see the same level of improvement. It also works best on recent injuries. However, it provides concrete, human proof that paralysis recovery is no longer an impossible dream. It shows that, with the right cells in the right environment, regeneration is possible. Why is Japan known for stem cell therapy for paralysis? Japan is known for stem cell therapy because of its advanced research, particularly in iPS cells (a Nobel Prize-winning Japanese discovery), and its unique regulatory system that allows for the fast-tracking of promising regenerative treatments. Japan's reputation as a stem cell hub isn't an accident. It's built on two key pillars: scientific innovation and supportive regulation. First, the innovation: the technology behind induced Pluripotent Stem Cells (iPS cells) was developed at Kyoto University, earning a Nobel Prize in 2012. This discovery allows scientists to take adult skin or blood cells and "reprogram" them into an embryonic-like state, from which they can become any cell type—including nerve cells. This bypasses the ethical debates around embryonic stem cells and is a cornerstone of Japanese research. Second, the regulation. In 2014, Japan passed new laws—the "Act on the Safety of Regenerative Medicine (ASRM)" and the "PMD Act"—to create a special, fast-track pathway for regenerative medicine. This system allows for "conditional approval" of treatments that show promising early data. This means that, unlike in the US or Europe where it can take 10-15 years, a promising therapy can be made available to patients in Japan much more quickly, provided they are monitored and data is collected. This has turned Japan into a "living laboratory" for regenerative medicine, attracting patients and researchers from all over the world. What is the legal status of stem cell therapy in Japan? Stem cell therapy is legal in Japan, operating under a specific regulatory framework. The "Act on the Safety of Regenerative Medicine" allows clinics to offer approved treatments, but many are still considered experimental and are not the same as fully approved drugs. This is a crucial point to understand. "Legal" doesn't mean "universally approved and covered by insurance." Japan's system is tiered. On one hand, you have fully approved medical products, like Stemirac, which has conditional approval for treating subacute spinal cord injuries. On the other hand, you have many private clinics that operate under the ASRM law. Under the ASRM, a clinic can submit a detailed treatment plan (e.g., "Use the patient's own fat-derived stem cells to treat osteoarthritis") to a government-certified committee. If the committee approves the plan based on its safety and rationale, the clinic is legally allowed to offer that treatment, even if it's still considered experimental. This is why you'll see a wide variety of stem cell therapies offered in Japan that may not be available elsewhere. It's a system designed to balance patient access with safety, but it places a lot of responsibility on the patient to research the specific clinic and treatment plan. It's also important to note that treatments using iPS cells (like the Keio University trial) are still largely in the clinical trial phase and not available commercially in private clinics. What's typically offered in clinics are treatments using Mesenchymal Stem Cells (MSCs) derived from a patient's own fat or bone marrow. What types of stem cells are used in Japan for paralysis? The two main types are induced Pluripotent Stem Cells (iPS cells), used in cutting-edge clinical trials, and Mesenchymal Stem Cells (MSCs), which are more commonly used in clinics and for the approved product "Stemirac." Understanding the "ingredients" is key. Not all stem cells are the same. In Japan, the treatments for paralysis generally involve two different types: Induced Pluripotent Stem Cells (iPS cells): This is the "high-tech" option. As mentioned, these are adult cells reprogrammed to be pluripotent (meaning they can become *any* cell). In the Keio trial, they were turned into neural precursor cells—the building blocks of the nervous system. The hope is that they will directly integrate into the spinal cord and become new, functional neurons. This is a highly complex, targeted approach primarily seen in major university hospitals and clinical trials. Mesenchymal Stem Cells (MSCs): This is the more common type used in clinics, including for the conditionally approved product Stemirac. MSCs are "adult" stem cells, typically harvested from the patient's own (autologous) bone marrow or fat tissue. Their main power isn't necessarily becoming new neurons. Instead, they act as the "paramedics" of the body. When infused, they home in on the injury, powerfully reduce inflammation, release a flood of protective growth factors, and help modulate the immune system. They create a healthy, supportive environment that allows the body's own repair mechanisms to work better. So, you can think of iPS cells as trying to rebuild the road, while MSCs are like the crew that cleans up the crash site, puts out the fires, and directs traffic so repair trucks can get through. What is the difference between iPS cells and MSCs for paralysis? iPS cells are used to create new nerve cells to directly replace damaged ones (a "regenerative" approach). MSCs are used to manage the injury site by reducing inflammation and releasing growth factors (a "reparative" and "supportive" approach). Let's dig a little deeper into this. The approach you might receive in Japan depends heavily on this difference. The iPS cell approach is one of direct replacement. The theory is that the paralysis is due to lost nerve cells, so the solution is to add new ones. This is a highly complex process, with risks like the cells not integrating correctly or, in early fears, forming tumors (a risk that has been greatly reduced with new techniques). This is the cutting edge and is mostly found in research trials for very specific patients, like those with recent injuries. The MSC approach is more about support and modulation. It's less about rebuilding the spinal cord from scratch and more about saving what's left. Most of the long-term damage from a spinal cord injury comes from the secondary cascade—swelling, inflammation, and cell death that spreads from the initial impact. MSCs are brilliant at stopping this secondary damage. By calming inflammation and protecting existing neurons, they can preserve function that would otherwise be lost. This is why Stemirac (which uses MSCs) is approved for *subacute* injuries—to stop that secondary damage wave in its tracks. What is the "Stemirac" treatment in Japan? Stemirac is a conditionally approved stem cell product in Japan for treating subacute spinal cord injuries. It uses the patient's own (autologous) bone marrow-derived mesenchymal stem cells (MSCs) delivered through an intravenous (IV) infusion. Stemirac is a big deal because it was one of the first stem cell products to receive any kind of government approval for spinal cord injury. It was developed by a company called Nipro and received conditional, time-limited approval in 2018. This approval was based on a small study of 13 patients. The process is specific: It is intended for patients with subacute SCI (generally within 14-40 days of injury). Doctors harvest bone marrow from the patient's hip. The MSCs are isolated from the marrow and grown in a lab for several weeks to get a very large dose (hundreds of millions of cells). This large dose is then infused back into the patient through a simple IV drip. The cells are thought to travel through the bloodstream, detect the "danger signals" from the spinal cord injury, and accumulate there to do their work. The approval was controversial because the trial was small and not double-blinded (the "gold standard"). However, supporters argue that for patients with no other options, this accelerated access is a humane and necessary step. What is the cost of stem cell therapy for paralysis in Japan? The cost of stem cell therapy for paralysis in Japan can vary dramatically, from $15,000 to over $50,000 USD. This depends on the type of cells, the number of treatments, the clinic, and the patient's specific condition. This is the question on everyone's mind, and unfortunately, the answer is complex. The cost of stem cell therapy is not a single, fixed price. It depends on many factors. Treatments in major university hospitals as part of a trial (like the Keio iPS cell study) may be covered by research funds, but these are extremely difficult to get into. For private clinics offering MSC treatments, you are paying out-of-pocket. The price range is wide, but here’s a general breakdown of what you might see. Please note these are *estimates* to give you an idea, not exact quotes. Treatment Type Estimated Cost Range (USD) What It Typically Includes Single IV Infusion (MSCs) $15,000 - $25,000 Consultation, cell harvesting (if autologous), lab processing, and a single high-dose IV infusion of MSCs. Multi-Session Program (MSCs) $25,000 - $60,000+ A comprehensive package that may include 3-5 infusions over several weeks, plus associated therapies like physiotherapy. Direct Injection (Clinical Trial) Varies (Often covered by trial) This is for treatments like the iPS cell trial. It's not a commercial product. Costs are for the surgery, hospital stay, and monitoring, which may be covered by the research grant or national health insurance if it's an approved trial. Stemirac (Approved Product) Covered by Japanese Insurance* *If you are a resident of Japan and meet the extremely specific criteria (e.g., subacute SCI), the product itself is conditionally covered. Foreigners would likely not have access to this coverage. These costs typically do *not* include travel, accommodation, or long-term follow-up care. It's a significant financial undertaking, which is why it's so important to have a clear consultation with a clinic beforehand. Does Japanese national insurance cover stem cell therapy for paralysis? Only in very specific cases. The approved product "Stemirac" has conditional insurance coverage for Japanese residents with subacute spinal cord injuries. Most other stem cell treatments in private clinics are not covered and must be paid out-of-pocket. This is a common point of confusion. The headlines about Stemirac being "covered by insurance" are true, but they apply to a very narrow slice of the population: Japanese citizens or residents who are part of the national health insurance system and who suffer a spinal cord injury, and are treated within the "subacute" window. For international patients, or for patients with chronic (old) injuries, this coverage does not apply. Almost all treatments offered to medical tourists in private clinics are self-pay. You should operate under the assumption that you will be responsible for 100% of the cost. How successful is stem cell therapy for paralysis in Japan? Success rates are still being studied and are not guaranteed. The famous Keio University iPS cell trial showed significant improvement in 2 out of 4 patients (a 50% rate in a tiny group). Other studies show more modest, but still meaningful, improvements in sensation or motor function. This is where we must be both hopeful and realistic. "Success" can mean different things. For one person, it might mean regaining the ability to breathe without a ventilator. For another, it might mean regaining bladder control. For another, it's walking. The Keio University trial is the most dramatic example, with a 50% success rate in its first four patients. But it's a tiny, tiny sample size. The 13-patient study for Stemirac also showed that 12 of the 13 patients improved by at least one grade on the ASIA impairment scale (a standard for measuring SCI). However, critics pointed out that some spontaneous recovery is expected in subacute injuries, and there was no placebo group to compare against. Most clinics will not—and should not—promise a "cure." What they are offering is a *chance* at improvement. The success depends heavily on: The type of injury: A complete severing of the spinal cord is much harder to treat than a contusion or partial injury. The age of the injury: Subacute (recent) injuries generally respond much better than chronic (old) injuries, as there is less scar tissue. The patient's health: Overall health and commitment to rehabilitation play a huge role. The treatment protocol: The type of cells, the dose, and the delivery method all matter. What does the stem cell therapy procedure for paralysis involve? The procedure varies. It can be a non-invasive IV infusion (like for Stemirac), where cells travel to the injury, or a more complex surgical procedure involving direct injection of cells into the spinal cord (like the iPS cell trials). Your experience in Japan would be very different depending on the treatment you receive. For an IV Infusion (Common in clinics with MSCs): Consultation & Harvest: You'll have an initial consultation, blood tests, and scans. If using your own (autologous) cells, there will be a procedure to harvest them, often a "mini-liposuction" to get fat tissue or a bone marrow draw. Cell Processing: The harvested tissue is sent to a lab, where the MSCs are isolated and cultured. This can take 2-4 weeks. Infusion: You'll return to the clinic for the infusion. This is often as simple as sitting in a chair with an IV in your arm for 30-60 minutes. You are monitored for a short time after and then are free to leave. Repeat: This process might be repeated several times over a number of weeks. For a Direct Injection (Common in clinical trials with iPS cells): Screening: This is an extensive process to see if you are a perfect match for the trial's criteria (e.g., must be 3 weeks post-injury, ASIA-A grade, etc.). Surgery: This is a major neurosurgical procedure. The surgical team will carefully expose the injured part of your spinal cord. Injection: Using micro-needles and advanced imaging, the surgeons inject millions of prepared neural stem cells directly into and around the injury site. Recovery & Rehab: You will have a significant hospital stay for recovery, followed by months or even years of intensive, monitored physiotherapy. You will also need to take immune-suppressing drugs to prevent your body from rejecting the new cells. Who is a good candidate for this treatment in Japan? The best candidates are often those with "subacute" spinal cord injuries (a few weeks to a few months old). However, clinics may also treat "chronic" injuries (older than 6 months), though expectations for recovery may be more modest. This is one of the most important factors. The "timing" of the injury is critical. Most of the dramatic success stories, and the approved treatments like Stemirac, are for subacute spinal cord injuries. This is the "sweet spot" after the initial swelling has gone down but before dense, impenetrable scar tissue has formed. In this window, the stem cells have the best chance of stopping secondary damage and promoting repair. What about chronic injuries (e.g., someone who has been in a wheelchair for 5 years)? This is a much tougher challenge. The scar tissue at the injury site is a major physical barrier, and the neural pathways have been dormant for a long time. Many clinics in Japan *will* treat chronic patients, usually with high-dose IV infusions of MSCs. The goal here is less about "rebuilding" the spine and more about "optimizing" it—reducing chronic inflammation, improving nerve signaling, and potentially waking up dormant pathways. Improvements are often more subtle: a recovery of some sensation, reduced pain, or slight improvements in motor control, rather than walking again. It's about improving quality of life. What is a "subacute" spinal cord injury and why does it matter? A "subacute" spinal cord injury is one that is no longer in the initial, acute phase (first few days) but is not yet chronic. This period, typically from 2 weeks to 6 months post-injury, is considered the prime window for stem cell intervention. Think of a spinal cord injury like a major car crash on a highway. Acute Phase (First 0-14 days): This is the crash itself. There's chaos, fire, and explosions (swelling, inflammation, cell death). It's too dangerous and chaotic to start repairs. The focus is on stabilization. Subacute Phase (2 weeks - 6 months): The fires are out, but the wreckage is still smoldering. This is the critical window. If you can get a cleanup crew (MSCs) in *now*, you can clear the debris, stop smoldering fires (inflammation), and prevent the whole highway from being permanently blocked. This is when Stemirac is used. Chronic Phase (6+ months): The wreckage has been left for so long that a giant, permanent concrete barrier (scar tissue) has been built across the highway. Now, just cleaning up isn't enough. You have to somehow break through that barrier, which is much, much harder. This is why all the most promising research and approved treatments target that subacute window. It's the moment of greatest opportunity for intervention. What are the risks or side effects of this therapy? The risks are generally considered low for MSC infusions, with the most common side effects being temporary headache, fever, or fatigue. The risks for surgical injection (iPS cell trials) are higher, as they include the risks of major spinal surgery and immune-suppressing drugs. No medical procedure is zero-risk, but the safety profile for MSC therapy is one of its biggest advantages. Because the cells are often your own (autologous), there is no risk of rejection. The Keio University iPS cell trial, which used donor cells, reported no serious adverse effects. The Mayo Clinic's similar trial in the U.S. also concluded the procedure was safe, with minor side effects like headaches. For MSC IV infusions, the risks are minimal and may include: Headache or fever, which usually resolves within 24 hours. Fatigue after the infusion. Risk of infection at the IV site (same as any IV). For iPS cell surgical injections, the risks are more significant because it's a major operation: Standard risks of anesthesia. Risk of infection at the surgical site. Risk of bleeding or spinal fluid leak. Risks from the immune-suppressing drugs (like high blood pressure or increased risk of infection), which are required for donor cells. An early fear with iPS cells was the risk of them forming tumors (teratomas). However, researchers have gotten much better at purifying the cells, and this risk is now considered very low and is watched for intensely in all trials. How long is the recovery period after treatment? For an IV infusion, there is virtually no recovery period. For surgical injections, hospital recovery is several weeks. However, the "recovery" in terms of neural function takes many months, or even years, and requires intensive, ongoing rehabilitation. This is a critical point: the stem cells are not a "fix." They are the *start* of a new recovery process. You don't get the infusion and walk the next day. The stem cells create the *potential* for repair, but the brain still needs to relearn how to use those new or repaired pathways. Think of the stem cells as planting a seed. You still have to water it, give it sunlight, and protect it. That "watering" is rehabilitation. Every successful stem cell study has been paired with intensive physiotherapy. The patient in the Keio trial who can now stand has been in active training to relearn how to walk. You must be prepared for a long-term, dedicated rehabilitation program to take advantage of any openings the stem cells create. What is the process for a foreigner to get stem cell therapy in Japan? Foreigners can get stem cell therapy in Japan by contacting private clinics or medical tourism facilitators. The process involves submitting medical records for review, an online consultation, and then traveling to Japan for the treatment, which is paid for out-of-pocket. Japan's system is open to international patients, but it requires careful planning. Research: This is the most important step. You need to find reputable clinics. Look for clinics that are transparent about their treatment, have clear pricing, and are registered with the Ministry of Health, Labour and Welfare (MHLW). Consultation: You will typically start with an online consultation. You (or a medical facilitator) will send all your medical records, MRIs, and a history of your injury. The clinic's doctors will review your case and determine if you are a candidate. Treatment Plan & Cost: If you are accepted, the clinic will propose a detailed treatment plan (e.g., "Three infusions of 200 million adipose-derived MSCs") and a clear cost estimate. Travel & Logistics: You will need to arrange a medical visa (if required), flights, and accessible accommodation. Many clinics or their partner agencies (like PlacidWay) can help with these logistics. Treatment: You will travel to Japan for the treatment, which could take anywhere from a few days for a single infusion to several weeks for a multi-session protocol. Follow-up: The clinic will provide follow-up instructions, and you will typically be monitored remotely after you return home. What should I look for in a Japanese stem cell clinic? Look for a clinic that is officially registered with the Japanese Ministry of Health, Labour and Welfare (MHLW). They should be transparent about their treatment plan, the source and number of cells, the risks, and the cost, and they should not make unrealistic promises of a "cure." This is your most important task as a patient. Because this field is so new, and there is so much hope, it can be a target for bad actors. Here's a checklist of what to look for: Government Registration: This is non-negotiable. Ask for proof that the clinic and its treatment plan are registered with the MHLW under the ASRM. Transparency: Do they clearly explain the *type* of cells (MSCs? iPS?), the *source* (your own fat? a donor?), and the *dose* (how many million cells)? If they are vague, this is a red flag. Realistic Claims: Be very wary of any clinic that "guarantees" results or promises a "cure for paralysis." Reputable doctors are hopeful but cautious. They will talk about "potential for improvement" and "quality of life." Specialization: Do they have experience with neurological conditions and spinal cord injuries, or do they just treat wrinkles and sore knees? Look for specialists. Clear Pricing: You should receive a detailed, itemized quote with no hidden fees. Good Communication: Do they have English-speaking staff? Do they answer your questions patiently and completely? You must be able-to communicate clearly about your health. Finding the right path to recovery can feel overwhelming, but you don't have to do it alone. The world of regenerative medicine is complex, but it's filled with new possibilities. If you're ready to explore your options for stem cell therapy in Japan or other leading destinations, PlacidWay is here to help. We connect patients with a global network of accredited clinics and hospitals. Let us help you get a free, no-obligation consultation and find the solution that's right for you.
Can Stem Cells Slow Parkinson’s Progression in Japan?
Recent clinical trials in Japan using induced pluripotent stem (iPS) cells have shown significant promise. This therapy has demonstrated it can be done safely and may slow Parkinson's progression by replacing lost dopamine-producing neurons, with some patients showing improved motor function. Hello! If you or a loved one are navigating the challenges of Parkinson's disease, you've likely heard whispers and hopeful reports about stem cell therapy, particularly from Japan. It's a topic filled with complex science but also immense hope. For decades, Parkinson's treatment has focused on managing symptoms, primarily replacing the brain's lost dopamine with medications. While these drugs are essential, they don't stop the underlying disease from progressing. This is where the groundbreaking work in Japan comes in, offering a completely different approach. Instead of just managing the deficit, scientists are working to *replace* the very cells that Parkinson's destroys. Japan has become a global leader in this field, largely due to the discovery of induced pluripotent stem cells (iPS cells), which earned a Nobel Prize. These are not embryonic stem cells; they are adult cells (like skin or blood) reprogrammed to become any cell in the body, including the specific dopamine-producing neurons that are lost in Parkinson's. Recent clinical trials from prestigious institutions like Kyoto University are moving this from theory to reality. They are not just asking if it's possible, but if it's safe and effective at slowing, or perhaps one day even reversing, the relentless progression of this disease. We're going to dive into exactly what this treatment is, what the latest 2025 findings show, how much it might cost, and what it could mean for patients around the world. This isn't about miracle cures, but about real, tangible scientific progress. We'll break down the common questions, separate the hype from the reality, and give you the clear, expert answers you're searching for. Let's explore the current landscape of stem cell therapy for Parkinson's in Japan together. What is stem cell therapy for Parkinson's disease? Stem cell therapy for Parkinson's is a regenerative treatment that aims to replace the dopamine-producing brain cells (neurons) destroyed by the disease. The goal is to restore the brain's ability to produce its own dopamine, thereby reducing symptoms and potentially slowing disease progression. Traditional Parkinson's treatments, like Levodopa, essentially provide the brain with the dopamine it's no longer making. This stem cell therapy is different; it's a form of regenerative medicine. The core idea is to transplant new, healthy cells—grown from stem cells—directly into the specific brain regions (like the putamen) where the original neurons have died off. Once transplanted, these new cells are intended to mature into functional dopamine-producing neurons. If successful, they integrate into the brain's existing circuitry, start producing dopamine, and restore the communication pathways that control movement. This is a fundamental repair strategy, which is why it holds the potential to not just mask symptoms but to create a lasting, biological change and slow progression. How does Parkinson's disease affect the brain? Parkinson's disease primarily affects a small area of the brain called the substantia nigra. In this area, nerve cells that create a vital chemical called dopamine begin to die off. This loss of dopamine disrupts signals to brain regions that control movement, leading to the classic motor symptoms of Parkinson's. Think of dopamine as a crucial messenger that allows for smooth, coordinated muscle movement. When you decide to walk, type, or smile, dopamine relays that signal efficiently. In a person with Parkinson's disease, the cells producing this messenger are progressively lost. As dopamine levels drop, the signals become weak and erratic. This disruption is what causes the hallmark symptoms: Tremor: Shaking, often beginning in a hand or finger. Bradykinesia: Slowness of movement, making simple tasks difficult. Rigidity: Stiffness in the limbs or trunk. Postural Instability: Problems with balance and coordination. Because the disease is progressive, this cell loss continues over time, and symptoms worsen. The goal of stem cell therapy is to directly replenish this specific population of lost cells. Can stem cell therapy really slow the progression of Parkinson's? Yes, evidence from recent clinical trials in Japan suggests it is possible. By transplanting new, healthy dopamine-producing cells, the therapy aims to restore the brain's depleted dopamine system. This could functionally halt or slow the progression of symptoms, as the new cells are not affected by the disease in the same way. This is the central question and the most exciting part of the research. The Kyoto University trial, with results published in 2025, provides the strongest evidence to date. Researchers observed that among the patients evaluated, several showed measurable improvements in their motor function scores even when they were off their standard medications. This is a key indicator that the treatment is having a genuine effect. Furthermore, brain scans (specifically PET scans) confirmed that the transplanted iPS cells were surviving, integrating, and—most importantly—producing dopamine two years after the procedure. This suggests the treatment can create a new, lasting source of dopamine. While no one is using the word "cure," this is the first therapeutic approach that has demonstrated a potential to create a long-term biological repair, fundamentally changing the disease's trajectory for a patient. What are iPS cells, and why are they used in Japan? Induced Pluripotent Stem Cells (iPS cells) are adult cells (like skin or blood) that are genetically reprogrammed back into an embryonic-like, "pluripotent" state. This means they can be coaxed into becoming any cell type, including the dopamine neurons needed for Parkinson's treatment. Japan is the birthplace of iPS cell technology, discovered by Dr. Shinya Yamanaka at Kyoto University. This discovery is a source of national pride and a focus of intensive government and academic research. Using iPS cells cleverly sidesteps the ethical and logistical hurdles of using embryonic stem cells. There are two main advantages: No Ethical-Sourcing Issues: Since they come from adult donors (or even the patient themselves, known as "autologous"), they avoid the controversy associated with embryos. Reduced Rejection Risk: The Kyoto trial used iPS cells from healthy donors whose immune profiles (HLA-matched) are compatible with a large portion of the Japanese population. This makes them "off-the-shelf" cells that are less likely to be rejected by the patient's immune system, reducing the need for heavy immunosuppressant drugs. Is stem cell therapy for Parkinson's approved in Japan? As of late 2025, there is no *fully* approved and commercially marketed stem cell therapy for Parkinson's in Japan. However, the system is unique. Some treatments are available under Japan's "Act on the Safety of Regenerative Medicine (ASRM)," which allows clinics to offer experimental therapies with regulatory oversight. This is a critical distinction. The groundbreaking iPS cell treatment from the Kyoto University trial is not yet available to the public. Sumitomo Pharma, the company that manufactured the cells, has filed for regulatory approval in Japan based on the trial's promising results. It is currently under a priority review, meaning a decision could come relatively soon. Separately, some private clinics in Japan offer other types of stem cell treatments (often using stem cells from fat or bone marrow) for Parkinson's. These treatments are offered under the ASRM framework, which means the government has accepted their treatment plan as meeting safety standards, but it does *not* mean the treatment has been proven effective. These are still considered experimental and are typically very expensive. What is the status of clinical trials for Parkinson's stem cell therapy in Japan? Clinical trials are active and producing very promising results. The most significant is the physician-led Phase I/II trial at Kyoto University Hospital, which transplanted iPS cell-derived dopamine neurons into seven patients. The results, published in April 2025, confirmed the treatment's safety and showed potential efficacy. This trial is the one generating global headlines. Its primary goal was to check for safety, and it passed with flying colors: no serious adverse events, no tumors, and no uncontrolled movements (dyskinesias) were reported over the two-year follow-up period. This safety profile is a massive achievement. The trial also looked at efficacy. As mentioned, four of the six evaluated patients showed improved motor function, and PET scans confirmed the cells were alive and working. Based on this success, the pharmaceutical company Sumitomo Pharma is also running trials in the U.S. and is seeking approval in Japan. This marks a major step from academic research toward a widely available, approved medical treatment. Is it safe to get stem cell therapy for Parkinson's in Japan? Safety depends heavily on the treatment type. The iPS cell clinical trials at Kyoto University have demonstrated a strong safety profile over two years. However, treatments at private, for-profit clinics operating under the ASRM are still experimental, and their long-term safety and efficacy are not as rigorously proven. The main concerns with any stem cell therapy, especially in the brain, are: Tumor Formation: The risk that transplanted stem cells could grow uncontrollably. The iPS cells used in the Kyoto trial are carefully differentiated into neurons *before* transplantation to minimize this risk, and no tumors were seen. Uncontrolled Movements (Dyskinesias): This was a major side effect in older trials using fetal tissue. The new iPS cell-derived neurons appear to be much safer, with no graft-induced dyskinesias reported. Rejection: The patient's immune system could attack the new cells. This is managed by using HLA-matched donor cells and a temporary course of immunosuppressant drugs. While Japan's top research institutions are world-class, patients must be cautious about "stem cell tourism" and clearly understand the difference between a rigorous clinical trial and an experimental treatment offered at a private clinic. How is the stem cell procedure for Parkinson's performed in Japan? In the advanced iPS cell clinical trials, the procedure is a form of neurosurgery. Using precise imaging, surgeons transplant millions (e.g., 5 to 10 million) of lab-grown dopamine-producing neurons directly into the putamen, a deep brain structure severely affected by Parkinson's. This is not a simple injection. It is a highly specialized surgical procedure. The cells are delivered using a very fine needle, guided by MRI, to ensure they are placed in the exact locations where they are needed most. The patient is under anesthesia, and the procedure is performed by a team of neurosurgeons and neurologists. Some private clinics, however, may offer different, less-proven methods, such as intravenous (IV) infusions or injections into the spinal fluid. These methods are not believed to be effective for Parkinson's disease because the stem cells are unlikely to cross the blood-brain barrier and turn into the specific neurons needed in the correct location. What is the cost of stem cell therapy for Parkinson's in Japan? The cost of stem cell therapy for Parkinson's in Japan varies dramatically. Treatments in formal clinical trials (like the Kyoto University study) are typically funded by the research body or government and are not a direct cost to the patient. However, experimental treatments at private clinics are very expensive, often ranging from $25,000 to $100,000 USD or more. These private clinic costs are paid out-of-pocket and are not covered by insurance, as the treatments are not yet approved as a standard of care. The price can depend on the type of stem cells used (e.g., adipose-derived), the number of cells, the number of infusions, and the clinic's reputation. It's vital to get a clear, itemized quote before considering such a treatment. Here is a general cost comparison for context, though prices for experimental treatments are not standardized: Treatment Type Location Estimated Cost (USD) Notes iPS Cell Clinical Trial Japan (e.g., Kyoto University) $0 (for patient) Funded by research. Not open to the public; strict eligibility. Adipose/Mesenchymal Stem Cell (MSC) Therapy Private Clinic (Japan) $25,000 - $80,000+ Experimental (ASRM regulated). Efficacy for Parkinson's is not well-proven. MSC Therapy Clinics in Other Countries (e.g., Panama, Mexico) $15,000 - $50,000 Regulatory standards vary widely. High risk of unproven treatments. Standard Parkinson's Medication (Annual) USA / Europe $2,500 - $10,000+ Ongoing cost for symptom management. Does not slow progression. Who is an eligible candidate for this treatment? Eligibility is extremely specific. For the iPS cell clinical trials in Japan, participants were highly selected. Criteria included being between 50-69 years old, having Parkinson's for at least five years, and still being responsive to standard dopamine medication. These strict criteria are used to ensure the trial can accurately measure safety and efficacy. Patients with very advanced Parkinson's or those who no longer respond to Levodopa were generally excluded. Some trials were also limited to residents of Japan. For private clinics offering experimental treatments, the eligibility criteria are often much looser. This may seem appealing, but it also reflects the less rigorous, non-standardized nature of the therapy being offered. Reputable providers will still require a thorough medical evaluation to rule out contraindications. What is the recovery process like? The recovery process involves two phases. First is the short-term recovery from the neurosurgery, which involves a hospital stay. Second is the long-term monitoring, which includes a temporary course of immunosuppressant drugs (for about 6-12 months) to prevent rejection of the new cells. This is not an "in-and-out" procedure. After the brain surgery, patients are monitored closely in the hospital for several days. Once discharged, they begin the immunosuppression regimen. This is a critical period, as these drugs lower the body's overall immune defense, increasing the risk of infection. Patients must be careful and have regular follow-up appointments. It's also important to manage expectations. The benefits are not instant. The transplanted cells need months, or even a year or more, to fully mature, integrate, and start producing a significant amount of dopamine. The Kyoto trial followed patients for two years to properly assess the long-term effects. What is the success rate of stem cell therapy for Parkinson's in Japan? It is too early to state a "success rate" in percentages. However, the results from the Kyoto iPS cell trial are a strong indicator of success. In that small study, four out of six evaluated patients showed measurable improvements in motor function, and PET scans confirmed cell survival and dopamine production in the brain. This is a remarkable outcome for a Phase I/II trial, which is primarily focused on safety. "Success" here is defined as: Safety: The treatment did not cause harm. (Achieved) Cell Survival: The transplanted cells lived. (Achieved, confirmed by scans) Efficacy: The cells produced dopamine and reduced symptoms. (Achieved in a majority of the small group) This is a "proof-of-concept" success. It shows the therapy *can* work. Larger Phase III trials will be needed to determine *how well* it works across a larger, more diverse population and to establish a true statistical success rate. What is the difference between iPS cell therapy and adult stem cell (e.g., adipose) therapy? iPS cell therapy for Parkinson's involves creating *new* dopamine-producing neurons in a lab and surgically transplanting them to replace what was lost. Adult stem cell (MSC) therapy, often using cells from fat (adipose) tissue, does not create new neurons. Instead, these cells are typically infused intravenously to reduce inflammation and provide supportive "growth factors," which may protect existing neurons. This is the most important difference for a patient to understand. The iPS cell approach is a *replacement* strategy. It's like planting new trees in a forest that burned down. The adult stem cell (Mesenchymal Stem Cell or MSC) approach is a *support* strategy. It's like adding fertilizer and water to the remaining trees to help them survive longer. While reducing inflammation (the support strategy) may be beneficial, only the iPS cell replacement strategy directly addresses the core problem of Parkinson's disease: the massive loss of dopamine-producing cells. This is why the scientific community is so focused on the iPS cell trials in Japan. How do I find a reputable clinic for stem cell therapy in Japan? Finding a reputable clinic means looking for transparency, a connection to formal research, and adherence to Japan's ASRM regulations. Be wary of clinics that make "cure" promises, lack published data, or downplay risks. The safest route is often through major university hospitals and research institutions. Here's what to look for: Affiliation: Is the clinic part of a major university hospital (like Kyoto University Hospital) or research institute? Transparency: Do they clearly state what *type* of stem cells are used (i.e., iPS cells vs. adipose)? Do they explain the exact procedure? Data: Can they provide data, ideally published in peer-reviewed journals, for their specific treatment? ASRM Approval: Do they clearly show their treatment plan is approved by Japan's Ministry of Health, Labour and Welfare under the ASRM? Realistic Claims: Reputable doctors will be very cautious with their language. They will say "experimental," "shows promise," or "may slow progression." They will not say "cure" or "reverse." Be extremely skeptical of any clinic that relies heavily on patient testimonials instead of scientific data or that pressures you into making a quick decision. This is a major medical procedure, not a simple commodity. What is Japan's ASRM (Act on the Safety of Regenerative Medicine)? The ASRM is a unique Japanese law passed in 2014 that creates a framework for offering regenerative medicine treatments (like stem cell therapy) outside of the traditional, lengthy clinical trial process. It allows certified clinics to offer experimental therapies to paying patients, as long as the treatment plan is submitted to and approved by a government committee. This law is why Japan has so many clinics offering these advanced treatments. However, ASRM approval is *not* the same as full marketing approval from the PMDA (Japan's FDA). The ASRM committee primarily judges the *safety* of the proposed treatment and the clinic's ability to perform it, not its *effectiveness*. This "fast-track" system is designed to accelerate innovation, but it places a greater burden on the patient to understand that they are paying for an experimental treatment, not a proven one. How long does the treatment take? The iPS cell transplant is a single neurosurgical procedure that takes several hours. However, the total commitment is much longer, including a hospital stay of several days, and a follow-up period of immunosuppression and monitoring that lasts at least one to two years. This is not a quick trip. Patients traveling for this treatment would need to plan for an extended stay in Japan for the initial procedure and recovery. They would also need to coordinate long-term follow-up care with their neurologists back home, in communication with the Japanese medical team. Will I have to stop my Parkinson's medication? No, you will not stop your existing Parkinson's medication. In the Kyoto trial, patients continued their standard Levodopa regimen. The goal of the stem cell therapy is to supplement and restore the brain's own dopamine production, which may eventually allow for a *reduction* in medication, but this is a long-term goal. In fact, the trial measured success by testing patients' motor function *off* their medication, demonstrating the new cells were providing a benefit independent of their normal drugs. Any adjustments to your medication would be made very slowly and carefully by your neurology team long after the procedure. What are the next steps for this research? The next steps are regulatory approval and larger, double-blind Phase III clinical trials. Sumitomo Pharma has already applied for manufacturing and marketing authorization in Japan. If approved, this would be the world's first "off-the-shelf" iPS cell therapy for Parkinson's disease. A successful Phase III trial would be the final step to confirm the treatment's efficacy and safety in a larger, more diverse group of patients. This would move the therapy from an experimental procedure to a new standard of care, paving the way for it to become available to patients worldwide. Ready to Explore Your Healthcare Options? Navigating advanced medical treatments like stem cell therapy can be complex. PlacidWay is here to help you find and compare high-quality, safe, and reputable healthcare solutions around the world. Explore our network of accredited clinics and find the expert care you deserve. Explore PlacidWay Today
What Kinds of Stem Cells are Used for Knees Treatment in Japan?
The main kinds of stem cells used for knee treatment in Japan are Mesenchymal Stem Cells (MSCs), most often sourced from the patient's own (autologous) adipose (fat) tissue or bone marrow. Dealing with chronic knee pain, especially from knee osteoarthritis, can be incredibly frustrating. It can feel like it slowly steals away your favorite activities, from hiking to simply playing with your kids. You've likely tried the usual routes—painkillers, physical therapy, maybe even steroid injections—but the relief is often temporary. This is where the field of regenerative medicine in Japan is offering new hope. Japan has become a global leader in this area, not just because of its technology, but because of its strong regulatory framework that prioritizes patient safety. So, what's all the buzz about? At the heart of this revolution are stem cells, your body's own master repair cells. When it comes to stem cells for knees treatment in Japan, the most common and well-studied types are Mesenchymal Stem Cells (MSCs). These powerful cells are typically taken from your own body, either from a small sample of fat (adipose tissue) or from bone marrow. This is called an "autologous" treatment. In some cases, donor (allogeneic) cells from sources like umbilical cords are also used. These cells are special because they can help reduce inflammation, signal your body to heal itself, and potentially even support the repair of damaged cartilage. This isn't science fiction. This is a real, regulated treatment option that's helping people regain their mobility. But as with any advanced medical treatment, you probably have a lot of questions. What are the different types? Is it safe? How much does it cost? What is the process actually like? We're going to dive in and answer all of those important questions, giving you the clear, direct information you need to understand this cutting-edge treatment. What kinds of stem cells are used for knee treatment in Japan? Japan primarily uses Mesenchymal Stem Cells (MSCs) for knee treatment. These are most commonly autologous (from the patient's own body) and sourced from either adipose (fat) tissue or bone marrow. When you start researching, you'll see a few different terms. Let's break them down. The vast majority of treatments for knee osteoarthritis use Mesenchymal Stem Cells (MSCs). These are adult stem cells that are known for their strong anti-inflammatory properties and their ability to secrete "growth factors"—signals that tell your body's tissues to start repairing themselves. The key difference lies in where these MSCs come from: Adipose-Derived Stem Cells (ADSCs): These are taken from your own fat tissue, usually from the abdomen or thigh. This is a very popular source because fat is easy to access with a simple mini-liposuction procedure and is incredibly rich in MSCs—much more so than bone marrow. Bone Marrow-Derived Stem Cells (BM-MSCs): These are taken from your bone marrow, typically from the back of your hip bone (iliac crest). This has been a traditional source for stem cells, though the harvesting procedure can be more uncomfortable than fat collection. Both of these are typically "autologous," meaning they use your own cells, which eliminates any risk of rejection or immune system reaction. Some clinics may also offer "allogeneic" treatments, which use cells from a healthy, screened donor (like from an umbilical cord), but autologous fat-derived cells are the most common for orthopedic use in Japan. What are Mesenchymal Stem Cells (MSCs)? Mesenchymal Stem Cells (MSCs) are adult stem cells found in tissues like fat, bone marrow, and umbilical cords. They are used in knee treatments because they can powerfully reduce inflammation, release growth factors to promote healing, and signal other cells to repair damaged tissue. Think of Mesenchymal Stem Cells as your body's own specialized "repair crew." They aren't the same as embryonic stem cells, which can turn into any cell in the body. Instead, MSCs are adult stem cells that are smarter and more focused on their job: managing and repairing their local environment. Their power in treating knee osteoarthritis comes from a few key abilities: They are powerfully anti-inflammatory: Much of the pain from arthritis comes from chronic inflammation. MSCs are experts at calming this inflammation down, which can lead to significant and rapid pain relief. They release growth factors: MSCs act like a general contractor, releasing a cocktail of signaling molecules (growth factors) that tell your body's existing cells to get to work repairing cartilage, managing fluid, and improving the overall joint environment. They can modulate the immune system: They help balance the local immune response, stopping it from attacking the joint tissue. It's important to know that the primary benefit isn't from the stem cells "turning into" new cartilage. While they have the *potential* to, their main benefit in knee treatment is this powerful signaling and anti-inflammatory effect. They essentially help the knee heal itself by creating a much healthier environment within the joint. What is the difference between autologous and allogeneic stem cells? Autologous stem cells are taken from your own body (e.g., your own fat or bone marrow). Allogeneic stem cells are taken from a healthy, screened donor (e.g., an umbilical cord). Most knee treatments in Japan use autologous cells to avoid any risk of rejection. These two terms are fundamental to understanding your treatment options. Autologous means "from oneself." In this type of treatment, the process involves harvesting your *own* cells. For knee treatments, this means a doctor will take a small sample of your fat or bone marrow. These cells are then processed (and sometimes cultured to grow more of them) before being injected back into your knee. The huge advantage here is safety: there is virtually zero risk of your body rejecting the cells or having an allergic or immune reaction, because they are 100% your own. Allogeneic means "from another." These are stem cells that come from a compatible, healthy donor. The most common source is from the umbilical cord blood or tissue of a healthy newborn, which is ethically collected after birth. These cells are "immunoprivileged," meaning they are less likely to cause an immune reaction than other types of donor cells. They are processed, screened, and stored in a lab. The main advantage is that it's an "off-the-shelf" treatment—there is no harvesting procedure needed from you. However, most top Japanese clinics for orthopedic issues prefer using autologous cells due to the unmatched safety profile. Why are adipose-derived stem cells (ADSCs) so common? Adipose-derived stem cells (ADSCs) are very common because fat tissue is extremely easy to access and contains a much higher concentration of Mesenchymal Stem Cells (MSCs)—up to 500 times more—than bone marrow does. The harvesting procedure is also minimally invasive and less painful. For a long time, bone marrow was the "gold standard" source for stem cells. However, in the last decade, science has shown that adipose (fat) tissue is an incredibly potent and abundant source of MSCs. This has made it the preferred choice for many leading regenerative medicine Japan clinics. Here’s why ADSCs are so popular: High Cell Count: Fat is packed with MSCs. Per volume, it can contain 100 to 500 times more regenerative cells than an equal amount of bone marrow. This means doctors can get a very high-quality, high-quantity dose of cells for your treatment. Easy Harvesting: A small amount of fat (often just 20-50cc) can be harvested with a simple, quick mini-liposuction procedure done under local anesthetic. It's far less invasive and more comfortable for the patient than a bone marrow draw. High Quality Cells: The stem cells found in fat are robust and high-quality. They also don't seem to decline in quantity as much with age, which is a significant factor when treating older patients with osteoarthritis. Is stem cell therapy for knees legal and safe in Japan? Yes, stem cell therapy is legal and highly regulated in Japan. In 2014, Japan passed the Act on the Safety of Regenerative Medicine (ASRM). This law requires all clinics to get approval from the Ministry of Health, Labour and Welfare (MHLW) for their specific treatment plan, ensuring high standards of safety and oversight. This is one of the most important reasons Japan is considered a world leader. Unlike in some countries where clinics can operate in a "gray area," Japan's government has taken a proactive, positive stance. The Act on the Safety of Regenerative Medicine (ASRM) provides a clear legal framework for clinics to offer these advanced treatments. Under this law, a clinic can't just decide to start offering stem cell therapy. They must submit a detailed treatment plan to a government-accredited committee. This plan outlines: Exactly what kind of cells they will use (e.g., autologous adipose-derived). How the cells will be collected, processed, and (if applicable) cultured. The condition they are treating (e.g., knee osteoarthritis). All safety protocols and patient monitoring procedures. Only after this plan is approved and registered with the Ministry of Health, Labour and Welfare (MHLW) can the clinic legally offer the treatment. This system ensures that any clinic you visit is held to a high standard of safety, uses certified cell processing centers, and is required to track and report patient outcomes. This oversight provides a level of patient safety and trust that is unmatched. How much does stem cell therapy for knees cost in Japan? The cost of stem cell therapy for knees in Japan typically ranges from $6,500 to $13,000 USD (approximately ¥1,000,000 to ¥2,000,000 JPY). The final price depends on whether one or both knees are treated and if the cells are cultured to increase their number. This is a key question for anyone considering treatment. It's important to know that stem cell therapy is considered an elective, private treatment and is not covered by standard insurance. The cost can vary significantly based on several factors, but here is a general breakdown of what you might expect at a reputable Japanese clinic. The main factors influencing the price are whether you are treating one or two knees, and the "type" of treatment. Some treatments use a simple, same-day concentrate (called Stromal Vascular Fraction, or SVF), while others involve "culturing" the cells, which means taking the initial sample and growing them into many millions of new cells in a lab over several weeks. Culturing is a more complex and expensive process but provides a much higher number of cells for the injection. Estimated Cost Comparison: Stem Cell Knee Treatment in Japan Treatment Type Condition Treated Estimated Cost (JPY) Estimated Cost (USD) Local Injection (One Knee) Osteoarthritis (Mild to Moderate) ¥1,000,000 - ¥1,500,000 $6,500 - $9,700 Local Injection (Both Knees) Osteoarthritis (Mild to Moderate) ¥1,400,000 - ¥2,000,000 $9,000 - $13,000 Systemic IV Infusion + Local Systemic Anti-Aging / Wellness ¥3,400,000 - ¥6,000,000+ $22,000 - $38,800+ *Note: Prices are estimates and can vary based on the clinic, the complexity of your case, and currency exchange rates. Systemic infusions are often for anti-aging or autoimmune conditions and are not typically the primary treatment for an isolated knee issue. What factors influence the final cost? The final cost is mainly affected by: 1) The source of the cells (fat vs. bone marrow), 2) Whether the cells are cultured to increase numbers (which is more expensive), 3) The number of joints being treated (one knee vs. both), and 4) The reputation and location of the clinic. When you receive a quote from a clinic, it's important to know what's included. The price isn't just for the injection. It covers a comprehensive, high-tech medical process: Consultation & Diagnostics: Initial specialist consultation, blood work, and imaging like X-rays or MRIs. Cell Harvesting: The medical procedure to collect your fat or bone marrow. Cell Processing/Culturing: This is a major factor. Processing the cells for a same-day (SVF) injection is cheaper. Culturing them over 3-4 weeks in a certified lab (a CPC, or Cell Processing Center) to get 50-100 million cells is more expensive but provides a more potent therapy. The Injection: The final procedure where the doctor injects the cells into your knee, often using ultrasound guidance for perfect placement. Follow-up: Any follow-up appointments to monitor your progress. Always ask for a detailed quote that outlines exactly what is and is not included. A good clinic will be transparent about its pricing. Who is a good candidate for stem cell knee treatment? The ideal candidate has mild to moderate knee osteoarthritis (Grade 2-3), is experiencing chronic pain, and wants to avoid or delay knee replacement surgery. It is less effective for "bone-on-bone" (Grade 4) arthritis, as there is little remaining cartilage. This therapy is a fantastic option, but it's not a miracle cure for every person. The best results are seen in patients who are in the "treatment gap"—their arthritis is too advanced for simple pain-killers, but not yet severe enough to warrant a total knee replacement. You may be a good candidate if you: Have been diagnosed with mild to moderate (Grade 2-3) osteoarthritis. Experience daily knee pain that limits your activities. Are not getting enough relief from other treatments like physical therapy or injections. Are motivated to delay or avoid the need for major joint replacement surgery. Are in general good health (no active cancer, no systemic infections). It's important to have realistic expectations. Stem cell therapy is generally *not* effective for "bone-on-bone" (Grade 4) osteoarthritis. At that stage, there isn't enough remaining joint structure for the cells to work with, and a knee replacement is often the only viable solution. A reputable doctor will be honest with you about your chances of success. What is the process for getting stem cell therapy in Japan? The process for autologous (cultured) stem cells involves 2-3 visits. Visit 1: Consultation, tests, and fat collection (a 1-hour procedure). Lab Phase: Your cells are cultured for 3-6 weeks. Visit 2: The cultured stem cells (millions) are injected into your knee. For an international patient, the process is streamlined. Let's assume you are getting the most common type: cultured autologous adipose-derived stem cells. Step 1: Remote Consultation. You will start by sending your medical records, including MRIs and X-rays, to the clinic. You will have a video consultation with the doctor to discuss your case and determine if you are a good candidate. Step 2: Visit 1 (First Trip to Japan - approx. 1 week). You will have an in-person consultation, physical exam, and final blood tests. You will have the fat harvesting procedure. This is a simple, 30-60 minute procedure done under local anesthetic. A tiny incision is made, and about 20ml (a small spoonful) of fat is collected from your abdomen. Your fat sample is then sent to a certified Cell Processing Center (CPC) to begin the culturing process. You can fly home after a day or two. Step 3: The Lab Phase (3-6 weeks). This is when you are back home. In the lab, technicians isolate your MSCs from the fat and grow them in a special, safe medium. They multiply over several weeks until they reach a target number, often between 50 million and 100 million cells. Step 4: Visit 2 (Second Trip to Japan - approx. 1 week). You return to the clinic. Your new, powerful stem cells are ready. The treatment itself is a simple injection. The doctor injects the cells directly into your knee joint. The entire appointment takes less than an hour. You can walk out of the clinic and typically fly home the next day. What is the recovery time after a stem cell knee injection? Recovery is very fast. You can walk out of the clinic immediately after the injection. You may have some minor discomfort or swelling for 2-3 days. You should rest and avoid heavy activity for the first 1-2 weeks, but you can return to normal daily life right away. This is one of the biggest advantages over surgery. There is no lengthy, painful rehabilitation. The injection itself is the final step. Day 1-3: You may feel some soreness, fullness, or mild swelling in the knee. This is normal. You'll be advised to take it easy, rest, and ice the knee. Week 1-2: You should avoid high-impact exercise (running, jumping) and heavy lifting. However, gentle walking and normal daily activities are perfectly fine. Week 3-4: You can usually resume light exercise, like swimming or cycling. Many clinics will also provide a gentle physical therapy plan to help you get the best results. After 1 Month: Most patients can return to all their usual activities. Improvement is not instant. The cells need time to work, reduce inflammation, and signal healing. Most patients start to notice significant improvements in pain and mobility around the 3-week mark, with continuous improvement over the next 3-6 months. What are the risks or side effects of stem cell therapy for knees? When using your own (autologous) cells, the risks are extremely low. The most common side effects are minor and temporary, such as pain, swelling, or bruising at the injection site or the fat collection site. The risk of infection is very low in a certified clinic. Because the treatment uses your body's own cells, it is exceptionally safe. The primary risks are the same as any needle-based procedure: Pain/Swelling: Temporary discomfort at the injection site in the knee or the (also temporary) soreness at the fat harvest site. Infection: This is a risk with any injection, but in Japan's highly regulated, sterile clinics, this risk is extremely rare. No Improvement: The biggest "risk" is not a safety one, but one of efficacy. There is a chance the treatment may not work for you, especially if your arthritis is very advanced. There is no risk of rejection, allergic reaction, or immune response when you use your own autologous cells. This is what makes it such a safe alternative to surgery or long-term medication use. What is the success rate of stem cell knee treatment in Japan? Success is defined by pain reduction and functional improvement, not by "regrowing" a new knee. Most clinical studies and patient reports from Japanese clinics show that 70-80% of patients with mild-to-moderate osteoarthritis experience a significant reduction in pain and an increase in mobility. It's crucial to define "success." We are not (yet) at the stage of "regrowing" a brand new cartilage cap, like a new tire on a car. The goal and the measured success of this treatment are about: 1. Significant pain reduction. 2. Improved function and mobility. 3. Delaying or eliminating the need for knee replacement surgery. By these measures, the success rate is very high. Most published data and clinic reports show that 70-80% of carefully selected patients (those with mild-to-moderate OA) see a major improvement in their quality of life. Many patients are able to get back to activities they thought they had lost for good. This is not a 100% guarantee. A small percentage of patients may not respond to the treatment. This is why the initial consultation with an honest doctor is so important, so they can give you a realistic assessment of your individual case. How is this different from PRP (Platelet-Rich Plasma) therapy? PRP (Platelet-Rich Plasma) uses concentrated platelets from your blood to release growth factors and signal healing. Stem cell therapy uses actual stem cells, which not only release growth factors but are also powerfully anti-inflammatory and can signal a much broader and more profound healing response. Stem cell therapy is considered a more powerful and advanced regenerative treatment. You may have heard of PRP, as it's more widely available and less expensive. Both are "regenerative" treatments, but they work differently. PRP Therapy: This involves drawing your blood, spinning it in a centrifuge to concentrate the platelets, and then injecting this "platelet-rich plasma" into your knee. Platelets are full of growth factors. Think of PRP as a "signal flare" that calls for healing. It's very good for mild arthritis or acute injuries like tendonitis. Stem Cell Therapy: This uses the stem cells themselves. Stem cells don't just send the signal; they *are* the "repair crew." They release a much wider and more sustained array of growth factors, are far more powerfully anti-inflammatory, and can manage the joint environment in a way PRP cannot. For moderate, chronic knee osteoarthritis, stem cell therapy is a much more powerful and comprehensive treatment. How do I choose a good stem cell clinic in Japan? To choose a good clinic, you must verify that it is legally registered with the Japanese Ministry of Health, Labour and Welfare (MHLW) under the ASRM law. Ask for their "Notification Number" and confirm they use a certified Cell Processing Center (CPC). This is your most important piece of homework. Any clinic can have a fancy website, but only legitimate clinics will have the proper government approvals. Here is your checklist: Ask for their MHLW Approval: This is non-negotiable. Ask for their "Notification Number" for their regenerative medicine plan. A reputable clinic will be proud to provide this. If they are vague, walk away. Confirm they use a certified CPC: Ask them *where* your cells will be processed. It should be in a certified "Cell Processing Center" (CPC) that meets strict government standards for safety and quality. Speak to the Doctor: You should have a consultation (even if remote) with the actual orthopedic doctor or specialist who will be treating you, not just a salesperson. Ask About Their Specialization: Do they specialize in orthopedics? A clinic that treats everything from "anti-aging" to "knees" may not have the same orthopedic expertise as a dedicated joint clinic. Look for Transparency: They should be open about costs, realistic about success rates, and honest about who is *not* a good candidate. Choosing a clinic that is properly regulated by the MHLW is your single greatest guarantee of safety and quality. Ready to explore your options for knee pain relief? Don't let knee pain hold you back. Discover the world-class regenerative medicine solutions available today. Explore top-tier, fully-regulated medical centers with PlacidWay.
When Will I See Results After Stem Cell Therapy for Hair Loss in Japan?
Most patients begin to see initial results after stem cell therapy for hair loss in Japan, such as reduced hair shedding, around 2 to 3 months. Significant, visible results in hair density and thickness typically appear between 6 and 12 months post-treatment. Watching your hair thin over time can be a deeply personal and frustrating experience. You've probably noticed more hair in the shower drain or on your brush and tried countless "solutions" that promise the world but deliver very little. It's a journey that can really impact your confidence. But in the field of hair restoration, there's a genuine shift happening, and Japan is at the forefront. We're moving beyond temporary fixes and into the realm of true regeneration. Stem cell therapy for hair loss in Japan is not a gimmick; it's an advanced medical treatment backed by a strong, safety-focused regulatory system. So, you're wondering, if I take this step, when will I *actually* see a difference? It's the most important question, and the answer is all about patience and understanding your body's natural hair growth cycle. Unlike a hair transplant, which moves hair, stem cell therapy works to *reactivate* your own dormant follicles. This process doesn't happen overnight. You can expect the first encouraging signs, like less shedding, to show up around the 2 to 3-month mark. The really exciting, visible changes in density—the kind you see in photos—start to become noticeable around 6 months and continue to improve for a full year or more. It's a process of healing and rebirth for your follicles. This guide will walk you through everything you need to know, from that all-important results timeline to the types of cells used, the cost, and what to expect from this cutting-edge hair regrowth results treatment in Japan. We'll answer all the questions you've been searching for, so you can make an informed and confident decision. What is stem cell therapy for hair loss? Stem cell therapy for hair loss is an advanced, non-surgical hair restoration treatment. It uses a patient's own (autologous) stem cells, typically from their fat tissue, to reactivate dormant hair follicles, reduce inflammation, and stimulate new hair growth in thinning areas. This treatment is a game-changer because it's *regenerative*. It's not a cover-up and it's not a transplant (like FUE or FUT), which simply moves follicles from one place to another. Instead, this therapy aims to fix the problem at its source: the weak, sleepy, or "miniaturized" hair follicles that are no longer producing healthy hair. The treatment involves injecting your scalp with a high concentration of your body's own master repair cells—specifically, Mesenchymal Stem Cells (MSCs). These cells are known as the "conductors" of your body's orchestra of healing. When placed in an area of thinning hair, they don't just sit there; they get to work, signaling your follicles to wake up and function properly again. It's about healing the scalp environment to support natural, healthy growth. How does stem cell therapy work for hair regrowth? Stem cell therapy works for hair regrowth by injecting millions of Mesenchymal Stem Cells (MSCs) into the scalp. These cells release a powerful cocktail of growth factors and anti-inflammatory signals that "wake up" dormant hair follicles, improve blood flow to the area, and reset the follicle's growth (anagen) phase. The science behind this is fascinating. Hair loss, particularly common androgenetic alopecia (male/female pattern baldness), is often caused by inflammation and a hormone called DHT, which makes follicles shrink. Stem cells fight this in several ways: Paracrine Signaling: The stem cells release hundreds of different growth factors and proteins (a "paracrine" effect) that act like a code, telling the shrunken follicles to restart the growth cycle. Anti-Inflammatory Action: They calm the chronic "micro-inflammation" around the follicles that contributes to hair loss. Angiogenesis: They promote the formation of new, tiny blood vessels, which increases the supply of oxygen and nutrients to the struggling follicles. Essentially, you are creating a much healthier, more fertile environment in your scalp, giving your existing (but dormant) follicles the best possible chance to start producing hair again. What types of stem cells are used for hair loss in Japan? The most common and effective type of stem cells used for hair loss in Japan are Adipose-Derived Stem Cells (ADSCs). These are Mesenchymal Stem Cells (MSCs) taken from a small sample of the patient's own (autologous) fat tissue, which is incredibly rich in these powerful regenerative cells. While stem cells can be found in bone marrow, the preferred source for most Japanese hair loss clinics is adipose (fat) tissue. There are a few key reasons for this: Higher Concentration: Fat tissue contains up to 500 times more MSCs per volume than bone marrow. This means a small, simple harvest can yield a very high and potent dose of cells. Easy Harvest: The "harvest" is a simple mini-liposuction procedure done under local anesthetic. It's minimally invasive, quick, and much more comfortable for the patient than a bone marrow draw. Some clinics may also offer treatments using allogeneic (donor) cells, such as from an umbilical cord bank. However, the standard for safety and efficacy in Japan is often autologous ADSCs, as using your own cells eliminates all risk of rejection or allergic reaction. When will I see results after stem cell therapy for hair loss in Japan? Patients typically see initial hair regrowth results from stem cell hair therapy in Japan within 2 to 3 months. This first sign is usually a reduction in hair shedding. More noticeable improvements in hair density and thickness become visible from 6 months onwards, with peak results often seen at the 12-month mark. This is the most critical question, and the answer lies in the human hair growth cycle. This cycle is naturally slow, and stem cell therapy works *with* it. The treatment "resets" the cycle for many dormant follicles, pushing them into a new "anagen" or growth phase. But that new hair has to grow from the root up. Here is a typical timeline of what to expect: Month 0-2 (The "Quiet" Phase): You've had the treatment. On the surface, nothing is happening. But beneath the skin, the stem cells are working, reducing inflammation and signaling follicles. The very first sign of success, often seen in month 2, is a significant decrease in hair shedding. Month 2-4 (The "First Fuzz" Phase): This is when the first new hairs, called vellus hairs, may begin to appear. They will likely be very fine, soft, and light in color, like "peach fuzz." This is a fantastic sign! It means the follicles are "on" again. Month 6-12 (The "Visible" Phase): This is when the magic happens. Those new vellus hairs, as well as other existing miniaturized hairs, begin to mature. They grow thicker, darker, and longer, becoming "terminal" hairs. This is when you and your barber or stylist will notice a real, visible increase in density and scalp coverage. This is why follow-up photos are so important. The change is so gradual that you might not notice it day-to-day, but a 6-month comparison photo can be dramatic. What do the first results look like? The very first results are often not new hair, but a significant reduction in hair shedding. You'll simply notice less hair in the shower drain or on your brush. This is followed by the appearance of fine, soft, "peach fuzz" (vellus) hairs in the treated areas. Managing expectations is key. You will not wake up a month after treatment with a full, thick head of hair. This is a biological process, not a cosmetic one. The first positive sign to look for is a stabilization of your hair loss. For many people, just stopping the shedding is a huge win. Following this stabilization, you'll enter the new growth phase. In addition to the new vellus hairs, you might also notice your *existing* hair looks healthier. The stem cells' growth factors can improve the quality and thickness of the "miniaturized" hairs you already have, making your hair look fuller even before the new growth fully matures. When will I see the *peak* results from the treatment? Peak hair regrowth results for stem cell therapy are typically observed between 12 and 18 months after the procedure. This extended timeline allows for multiple hair growth cycles to complete, giving new hairs time to mature from vellus hairs into strong, thick, terminal hairs. The stem cells themselves don't live in the scalp forever, but the healing cascade they initiate continues for a very long time. The cells release their growth factors, and your body's own repair systems continue the work. Because hair grows slowly (about 1/2 inch per month), it simply takes that long for the full effect of the "rebooted" follicles to become visible to the naked eye. This is why clinics in Japan will schedule follow-up appointments at 6, 12, and even 18 months. They are tracking this long-term improvement. Your 12-month photo will almost always look significantly better than your 6-month photo. How many stem cell treatment sessions are needed for hair loss? This depends on the clinic and the patient's condition. Some advanced Japanese hair loss clinics use high-dose, cultured stem cells, which often requires only one main treatment session. Other protocols may recommend a series of 2-3 smaller injections spaced a few months apart. There are two main approaches: High-Dose Cultured Cells: This is an advanced technique common in Japan. After your fat harvest, your cells are sent to a certified lab (CPC) and are "cultured" or grown for several weeks. This multiplies your 5-10 million harvested cells into 50 million, 100 million, or more. You then return for *one* very high-dose injection. This is often a "one and done" approach (for several years, at least). Non-Cultured Cells (SVF): This is a same-day procedure where the harvested fat is processed in a machine to isolate the stem cells (called Stromal Vascular Fraction, or SVF). This is a lower dose of cells, and some doctors may recommend a series of treatments to achieve the desired result. In either case, because hair loss is an ongoing genetic condition, your doctor may recommend a single "maintenance" treatment every 2-5 years to keep the results fresh. Is stem cell therapy for hair loss in Japan safe and legal? Yes, stem cell therapy for hair loss is legal and highly regulated in Japan. In 2014, Japan passed the "Act on the Safety of Regenerative Medicine" (ASRM). This law ensures that all clinics are approved by the Ministry of Health, Labour and Welfare (MHLW), guaranteeing high standards of safety, cell processing, and patient oversight. This is precisely *why* Japan is a top destination for this treatment. Unlike in many countries where clinics can operate in a regulatory gray area, the Japanese government has a clear, legal framework. This framework (the ASRM) requires any clinic offering stem cell therapies to: Furthermore, when using autologous (your own) cells, the treatment is exceptionally safe. There is no risk of rejection, immune reaction, or allergic response. It is your own body's healing mechanism, simply concentrated and redirected. What is the cost of stem cell therapy for hair loss in Japan? The cost of stem cell therapy for hair loss in Japan typically ranges from $4,000 to $10,000 USD (approximately ¥600,000 to ¥1,500,000 JPY). The price varies significantly depending on the source of the cells, whether they are cultured (a more complex process), and the clinic's protocol. This treatment is considered an elective procedure and is not covered by national health insurance. The price reflects the highly advanced technology, the cost of the certified lab processing, and the expertise of the specialists. Here is a general breakdown of what you might expect. "SVF" refers to the non-cultured, same-day procedure, while "Cultured ADSCs" refers to the more advanced, high-dose treatment that requires two visits. Estimated Cost Comparison: Stem Cell Hair Loss Treatment in Japan Treatment Type Estimated Cost (JPY) Estimated Cost (USD) Notes Single Treatment (SVF, non-cultured) ¥600,000 - ¥900,000 $4,000 - $6,000 Same-day procedure. Lower cell count. Single Treatment (Cultured ADSCs) ¥1,000,000 - ¥1,500,000 $6,500 - $10,000 High cell count (e.g., 50M+ cells). Requires 2 visits. Multi-Session Package (SVF) ¥1,200,000 - ¥1,800,000 $8,000 - $12,000 A series of 2-3 injections over several months. *Note: Prices are estimates and can vary based on the clinic, the size of the treatment area, and currency exchange rates. What factors influence the final cost? The main factors influencing the final cost are: 1) Cell Culturing (growing cells to a high number in a lab is more expensive than a same-day procedure), 2) The number of cells used (a 100-million cell dose costs more than 20 million), 3) The number of sessions, and 4) The clinic's reputation and technology. When you get a quote, it's not just for the injection. The price covers a comprehensive medical service, including: While a same-day SVF procedure is cheaper, many specialists in Japan prefer the cultured cell method as it delivers a much more potent, high-count dose, which may lead to more significant hair regrowth results. Who is a good candidate for this treatment? The best candidates are individuals in the early to moderate stages of hair loss (e.g., Norwood scale 2-5 for men, Ludwig scale 1-2 for women). This treatment works best when there are still "miniaturized" or dormant follicles that can be reactivated. This therapy is ideal for people who: Submit their entire treatment plan to a government-accredited committee for approval. Detail the cell source, processing method, and safety protocols. Use only certified, government-inspected Cell Processing Centers (CPCs). Track patient outcomes and report all data, including any adverse effects. The specialist consultation. The mini-liposuction (fat harvest) procedure. The complex, sterile lab work at the Cell Processing Center (CPC). The injection procedure, often done by a skilled doctor. All follow-up appointments. Are just beginning to see significant thinning and want to be proactive. Have "diffuse thinning," where the hair is weaker all over, rather than in one bald spot. Still have hair, even if it's very fine (this means the follicles are "asleep," not "dead"). Want a non-surgical, natural hair restoration option with no downtime. Are in general good health. Who is *not* a good candidate? This treatment is not effective for individuals with "slick bald" areas (e.g., Norwood 6-7), where the hair follicles have been completely lost for many years. It also does not work for scarring (cicatricial) alopecia or hair loss from active autoimmune diseases like alopecia areata. It's crucial to be realistic. Stem cells can "wake up" a sleepy follicle, but they cannot create a new follicle from scratch or revive one that is "dead" and gone. A reputable doctor will be honest with you. If your scalp is smooth and shiny, with no fine hairs visible at all, the follicles are likely gone, and a hair transplant (FUE) would be a more appropriate discussion. What is the treatment process like in Japan? For the advanced cultured stem cell method, it is a two-visit process. Visit 1 (1-2 days): This involves your consultation and the simple, 30-minute fat harvest procedure. You can then fly home. Visit 2 (3-6 weeks later): You return for the final treatment, which is a series of simple injections of your cultured cells into the scalp. Step 1: Consultation & Harvest (Visit 1). You'll meet the doctor and confirm you're a candidate. Then, under local anesthetic, a small amount of fat (about 20cc) is taken from your abdomen or flank. It's a quick procedure with no stitches. Your sample is then sent to the lab. Step 2: Cell Culturing (The "Wait"). For the next 3-6 weeks, you are back home. In the lab, technicians isolate your MSCs and grow them until they have a high-purity, high-count dose of millions of new cells. Step 3: Injection (Visit 2). You return to Japan. The doctor takes your new, powerful cells and injects them with a very fine needle into the areas of thinning on your scalp. The process is quick (about 30-60 minutes) and relatively painless (local anesthetic or numbing cream is used). You can walk out of the clinic and go to dinner. There is no downtime. What is the recovery time after a stem cell hair treatment? There is virtually no downtime. You can return to work and most normal activities immediately. You may have some minor scalp tenderness, redness, or swelling for 24-48 hours, but this is mild. You'll be advised to avoid heavy exercise or washing your hair for one day. This is one of the biggest advantages over surgical options. You don't have to "hide" for a week. The fat harvest site is tiny and just requires a small bandage. The scalp injections are done with such a fine needle that they are unnoticeable. You can fly home the day after your procedure. Are the results of stem cell hair therapy permanent? The hair regrowth results are long-lasting but not considered "permanent." The treatment does not stop the underlying genetic or hormonal cause of hair loss. A single treatment can show results for several years (e.g., 2-5 years), but your doctor may recommend a smaller "maintenance" session to maintain the results. Think of it like tending a garden. The stem cell therapy is like tilling the soil and adding a powerful, rich fertilizer, causing a beautiful bloom. But the underlying conditions (your genetics) are still there. Over time, the effects can fade as the aging and hair loss process continues. The good news is, a single, powerful treatment can "reset the clock" for years, and a simple maintenance injection can keep the results going strong. How does this compare to PRP (Platelet-Rich Plasma) therapy? Stem cell therapy is a more powerful and advanced treatment. PRP (from your blood) uses platelets, which are "signal flares" that release growth factors. Stem cell therapy (from your fat) uses the actual Mesenchymal Stem Cells (MSCs), which are the "master repair cells." These MSCs are far more anti-inflammatory and release a much wider, more potent range of growth factors for a longer period. PRP is a great, less-expensive option, but it often requires 3-5 sessions to see results. Stem cell therapy, especially the cultured cell method, is a much more robust, "heavy-duty" intervention. Many people see PRP as a good "maintenance" therapy after a more powerful stem cell treatment. How does it compare to a hair transplant (FUE/FUT)? They are completely different treatments. A hair transplant (FUE/FUT) is a surgical procedure that moves hair follicles from the back of your head (the donor area) to the front, bald area. Stem cell therapy is a non-surgical, regenerative treatment that reactivates your *existing* dormant follicles in the thinning area. You can't transplant hair to an area that is just "thinning." You need to shave the area. This is where stem cells shine—they are perfect for "diffuse thinning" to increase density *without* surgery. In fact, the two treatments work incredibly well together. Many of the world's best surgeons now use stem cell therapy *in combination* with a transplant. They will transplant hair to the bald spots and use stem cell injections in the thinning areas *behind* the transplant to increase overall density and protect the native hair. This is considered the "gold standard" for a complete hair restoration. What are the side effects of stem cell therapy for hair? Side effects are extremely rare, mild, and temporary, especially when using your own (autologous) cells. The most common side effects are temporary soreness, slight swelling, or redness at the injection sites on the scalp or at the small fat harvest site. Because you are using your own tissue, there is no risk of rejection, allergy, or transmitting disease. The entire process is "autologous." The main risks are the same as any needle-based procedure: a tiny risk of bruising or infection. In a high-end, MHLW-regulated Japanese clinic, this risk is negligible as they operate under the strictest sterile standards, just like a hospital. How do I choose a reputable hair loss clinic in Japan? You must choose a clinic that is legally registered with the Japanese Ministry of Health, Labour and Welfare (MHLW) under the ASRM (Act on the Safety of Regenerative Medicine). Always ask for their "Notification Number" and confirm they use a certified Cell Processing Center (CPC). This is the most important step you can take. Any clinic can have a flashy website, but only the best are compliant with Japan's strict laws. Here is your checklist: Ask for their MHLW approval. If they can't provide it, walk away. Ask where the cells are processed. It *must* be a certified CPC lab, not a machine in the office back room. Look for transparency. They should be honest about the cost, the timeline, and who is *not* a good candidate. Check their specialty. Look for a clinic that specializes in hair restoration and regenerative orthopedics, not just a general anti-aging clinic. Ready to take the next step in your hair restoration journey? Don't let hair loss define you. Discover the world-class regenerative medicine solutions available in Japan. Explore top-tier, MHLW-regulated medical centers with PlacidWay
在日本选择合适的干细胞治疗专家是获得世界最先进生物疗法的关键一步。我们精心挑选了东京、大阪及其他地区经验丰富的医生,他们在日本干细胞和综合再生医学领域享有盛誉。这些医生均经过严格评估,评估标准包括他们是否遵守日本的安全法规、临床研究以及患者疗效,从而帮助您找到最适合您再生医学需求的专家。
认证再生医学专家
已核实符合监管要求
在日本提供再生疗法的医生均严格遵守厚生劳动省的指导方针。他们的资质证明其接受过自体和异体材料处理方面的先进培训,使患者在接受高标准的医疗监督和符合伦理规范的治疗时,能够安心地在日本接受干细胞疗法。
先进的细胞培养技术
最先进的处理技术
日本许多提供先进细胞疗法的专家都利用世界一流的细胞处理中心(CPC)设施,以确保干细胞的纯度和效力。这些专家专注于精准的细胞分离和扩增技术,确保每一项再生医学方案都能最大限度地发挥治疗潜力并保障患者安全。
个性化再生方案
个性化生物解决方案
日本干细胞治疗领域的顶尖医生强调个体化治疗方案,根据骨科、神经科或抗衰老等特定目标制定治疗计划。从最初的细胞采集到最终的治疗,日本的再生疗法专注于每位患者的长期细胞健康和功能改善。
全球再生医学领导者
凭借开创性的研究和健全的监管体系,日本已成为全球领先的干细胞治疗中心。该国的医疗机构处于再生医学领域的前沿,为患者提供针对各种退行性疾病和慢性疾病的突破性治疗方案。
在日本寻求干细胞治疗的患者受益于日本以安全性和创新性为优先的医疗体系。通过将先进的细胞疗法融入临床实践,日本医院展现出严谨的科学性和精湛的技术,吸引着来自世界各地的患者。
创新生物治疗方案
选择日本的主要优势之一是日本拥有专业的再生疗法。这些疗法专注于利用人体自身的愈合能力修复受损组织和恢复器官功能,通常可以替代侵入性更强的传统手术。
无论是治疗骨科损伤、自身免疫性疾病还是延缓衰老,日本的干细胞疗法都采用高纯度细胞处理技术。这种对精准性的执着追求,确保了日本再生医学始终是患者改善生活质量的高效且备受青睐的解决方案。
高标准的临床安全性
日本先进细胞疗法的应用受到国家卫生部门的严格监管,确保每家诊所都遵守严格的质量控制规程。这种监管保证了日本干细胞均在符合最高国际生物安全标准的认证实验室中进行处理。
除了卓越的医疗水平,日本还提供有利于康复的环境,其特色在于世界一流的待客之道和高效的基础设施。对于国际患者而言,在日本接受再生疗法,意味着在一个以其医疗诚信和科技实力而闻名的国家,获得安全可靠的先进康复途径。
选择出国治疗是一项重大决定。PlacidWay 作为您值得信赖的合作伙伴,为您对接经过严格筛选的日本顶尖干细胞治疗诊所,确保您的康复之旅安全、透明、顺畅。
严格审查
我们会核实专家资质和医院认证,让您安心预订日本高级细胞疗法,因为您知道自己得到了专家的照顾。
定制报价
获取多家日本顶级再生医学诊所的详细免费报价。我们帮您比较不同套餐,确保您获得最佳性价比,且无任何隐性费用。
视频咨询
我们提供与专家的直接视频通话,让您在前往日本之前,可以讨论您的干细胞治疗目标并评估他们的沟通方式。
旅行资助
我们会协助您协调抵达、酒店预订和接送安排,作为您在日本进行再生疗法之旅的当地后勤指导。
患者权益倡导
从您最初的咨询到治疗后的随访,我们的团队随时为您提供支持,并确保您在住院期间与诊所进行有效的沟通。
已验证评论
查看以往患者的真实评价和成功案例,以验证治疗效果,并为您的日本先进细胞疗法之旅设定合理的预期。
立即体验未来医疗。联系 PlacidWay 获取免费的个性化报价,开始规划您在日本接受干细胞疗法的健康之旅。
获取您的免费个性化报价






.png)


















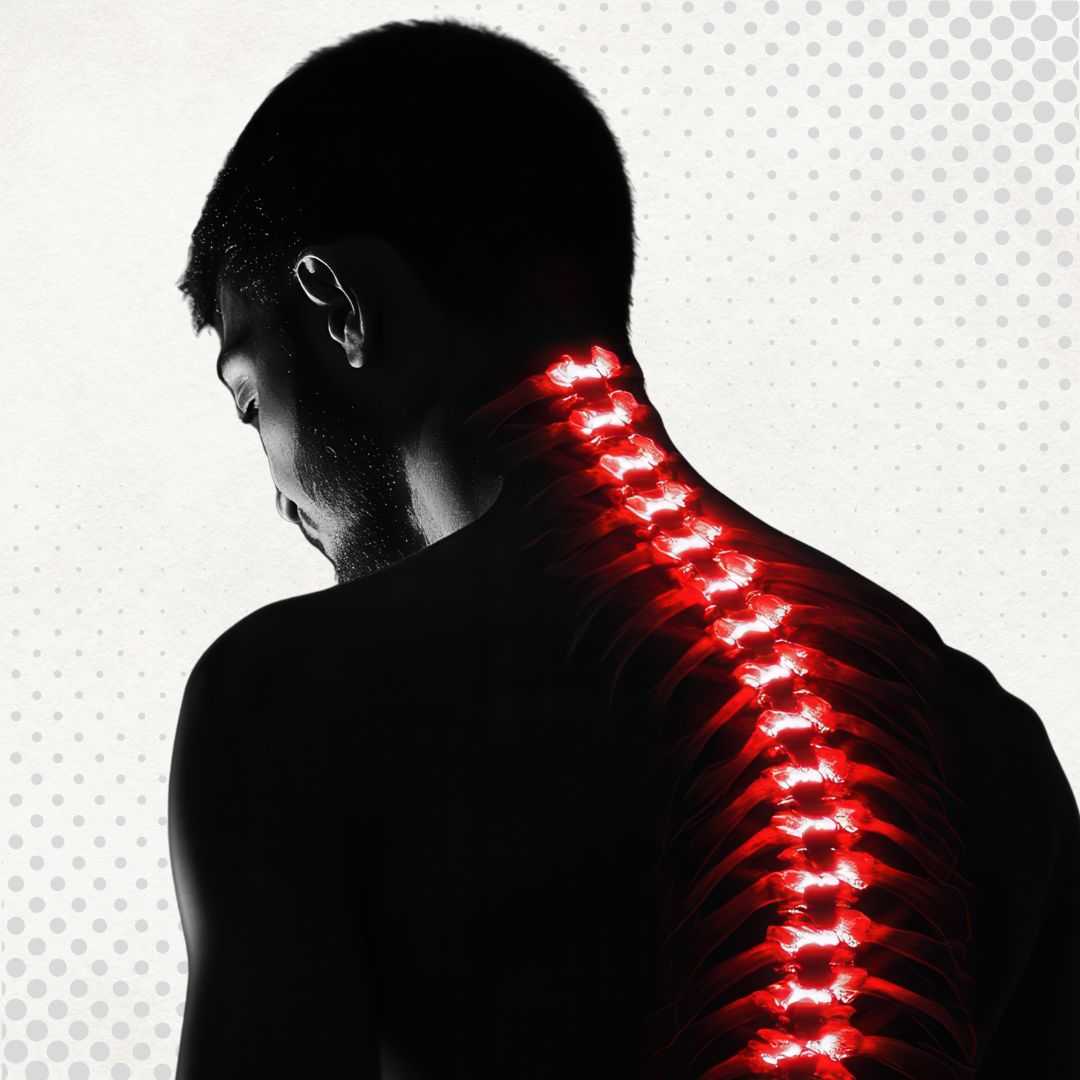




.png)
.png)
.png)


Share this listing